Transforming a Lab-Based Prototype into a Prototype for Clinical Feasibility
Hypoxia is a silent killer in critical care, often going undetected until it is too late. Early identification of oxygen deprivation at the cellular level could mean the difference between life and death.
Opticyte, a medical device startup spun out of the University of Washington, envisioned a groundbreaking real-time Cell O₂ monitor to address this challenge. However, their initial prototype was a complex, lab-based instrument—too large, too expensive, and impractical for clinical deployment. They needed to transition their promising technology from the lab to the bedside, but doing so required expertise in optical systems, human factors engineering, and scalable manufacturing. To meet this challenge, Opticyte partnered with StarFish Medical.

“I really value the team at StarFish.”
Lori Arakaki, Co-founder and CEO, Opticyte
Engineering Toward a Scalable, Clinically Focused Prototype
From the outset, StarFish Medical understood that Opticyte’s vision extended far beyond the research lab—it was about empowering clinicians with a practical tool that had the potential to meaningfully impact patient care. Achieving this required a fundamental redesign of both the hardware and sensor system, balancing performance, cost-effectiveness, and usability in what would be a high-stakes clinical environment.
StarFish engineers took a multi-pronged approach, tackling the project from several critical angles. First, they applied advanced optical engineering and precision manufacturing techniques to shrink the device from a bulky, lab-bound instrument into a streamlined bedside monitor. This miniaturization was a critical step toward enabling future clinical use, demonstrating how the technology might one day be integrated into critical care workflows.
The development team also had to rethink how the device interacted with patients. Traditional sensors were impractical for continuous use in dynamic ICU environments, where every second counts. To address this, StarFish designed an innovative adhesive sensor system that could be easily applied and removed, allowing for quick repositioning without compromising accuracy. This small but critical detail made the difference between a technology that worked in theory and one that could function reliably in the fast-paced world of emergency medicine.
Beyond hardware, the team at StarFish provided critical support in human factors engineering. Although the system was not intended to be a market-ready medical device, these early-stage considerations ensured the prototype could be used in a clinical research setting to evaluate feasibility and usability. StarFish’s engineering contributions laid a foundation for future development of features such as live algorithm processing, a user interface, and clinical alarms—all essential components of a commercial device but outside the scope of this prototype phase.
Delivering a Platform for Clinical Exploration
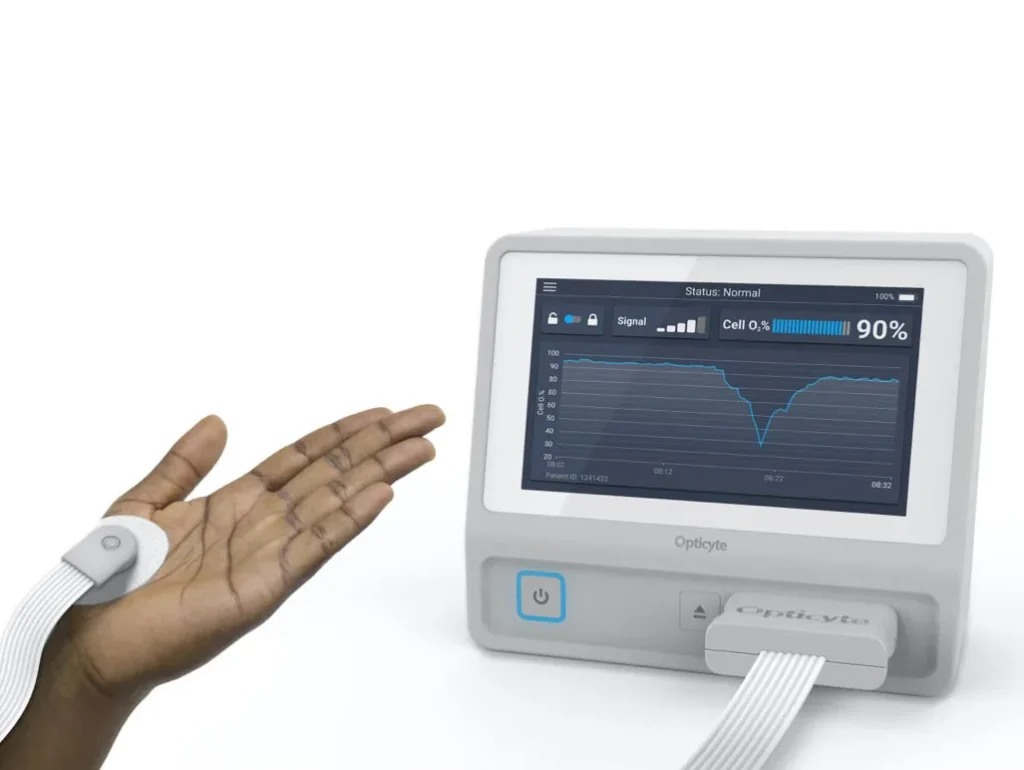
With StarFish Medical’s expertise, Opticyte developed an investigational prototype from its proof-of-concept lab instrument, capable of supporting early-stage clinical data collection. The collaboration delivered tangible results that go beyond technical specifications—it enabled Opticyte to take a meaningful step forward in its mission to revolutionize hypoxia detection.
- A Compact, Affordable Prototype: StarFish replaced an $80K spectrometer with a $4K optical subsystem, demonstrating that the core technology could be miniaturized and cost-reduced without compromising signal integrity—an essential step in proving feasibility.
- Designed with Clinical Context in Mind: The prototype’s sensor and system design enabled data collection in a research setting, reflecting the practical realities of potential clinical use without yet being a regulated device.
- Capital Efficiency and Accelerated Progress: By partnering with StarFish, Opticyte advanced their technology and collected early-stage data, fueling development and investor confidence while maintaining a lean internal team.
“One of my biggest concerns about hiring a third-party firm to do our engineering work was losing control over timelines and progress,” said Lori Arakaki, co-founder and CEO of Opticyte. “Having StarFish as our engineering partner instead of hiring our own team has been a wonderful solution. Our program manager understands both the technical and business needs of a startup, and we’ve made tremendous progress with limited capital.”
Laying the Groundwork for Future Medical Innovation
This collaboration is a testament to how StarFish Medical supports innovators in bridging the gap between breakthrough ideas and practical clinical exploration. For Opticyte, the outcome was a robust investigational platform that generated essential feasibility data and de-risked future product development.
With deep optical and systems engineering expertise, StarFish helped Opticyte move from lab vision to early clinical feasibility—providing the tools and technical foundation needed to shape their next phase of innovation.
