Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

In medical device development, verification is both a safeguard and a stress test, not just for the product, but for the process.
-

In the world of medical device development, requirements are often treated as a regulatory tax, essentially documentation created solely to satisfy a compliance need.
-

In this Bio Break episode, Nick and Nigel explore a surprising and memorable microbiology fact that puts everyday hand hygiene into perspective.
-

Nick and Nigel explore how a surprisingly small set of sensors could be used to identify a wide range of common health conditions.
-

Understanding how clinical ventilator development differs from commercial ventilator design is essential for teams planning early studies.
-

Nick walks through a practical Teflon tape lesson that came from real work supporting a mechanical test rig.
-
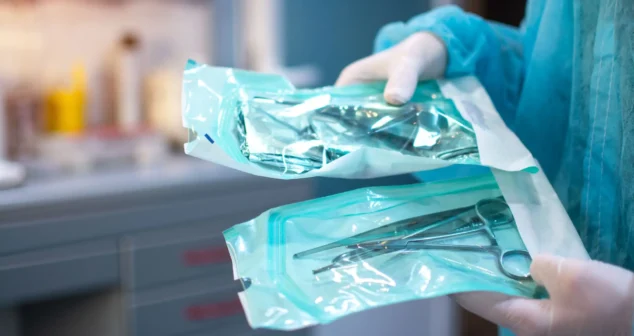
Most sterile medical devices begin their journey long before anyone thinks about sterilization. Teams focus on function, usability, materials, and suppliers, then discover that sterilization constraints can reshape many of those early decisions.
-

After years of working with founders and technical teams, I have learned that early design missteps rarely come from engineering flaws. More often than not, they come from missing conversations.
-

Medtech founders operate with more constraints than most sectors. You are responsible for deep technical problem solving, high-stakes decisions, regulatory navigation, investor conversations, and a constant stream of operational tasks.