Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

Closing the medtech skills gap: Who better to teach medtech skills within academia than those who are earning a living with those very skills?
-

Medical device development experts share their biggest medical commercialization lessons learned over 20 years.
-

Michael May, President and Chief Executive Officer of CCRM discusses the intersection of medical devices in Regenerative Medicine.
-

7 interesting outcomes of the 2019 Health Canada Action Plan on Medical Devices initiatives and alignment with international regulators.
-

9 medical device commercialization resolutions recommended by our team of engineers, designers, QA/RA, and manufacturing professionals.
-
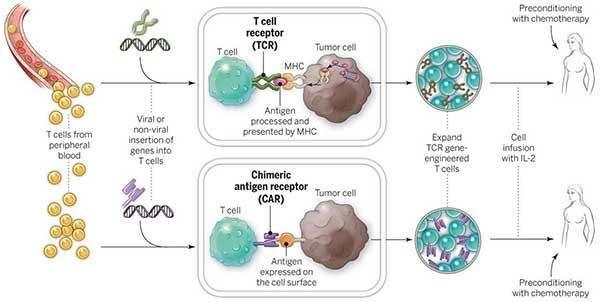
Chimeric antigen receptor T-cell (CAR-T) therapy is a rising shining star in Regenerative Medicine. Medical devices will be involved.
-

Advice to help companies determine when to start thinking about setting up a Startup Quality Management System.
-
Watch the most viewed StarFish Medical videos of 2018 to learn about medical device design, development, regulations, and manufacture.
-

New medtech blogs and new authors join our 2018 “most read” list. Regulatory was the most popular topic, followed by Electrical Engineering.