Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

Agile in medical device development improves quality, usability and effectiveness. Six considerations when implementing Agile.
-

Project Manager analysis of medical device development client-vendor perception gaps on cost, timeline, quality, and communication.
-

Results of StarFish client satisfaction survey with a large group that included customers and prospects using one word descriptions.
-

Efficiently integrating a distributed team for medical device launch is paramount to success. Use six tips outlined in the article.
-

Six ways to manage and communicate medical device project relationships without losing the essence of the development innovation or wasting money.
-
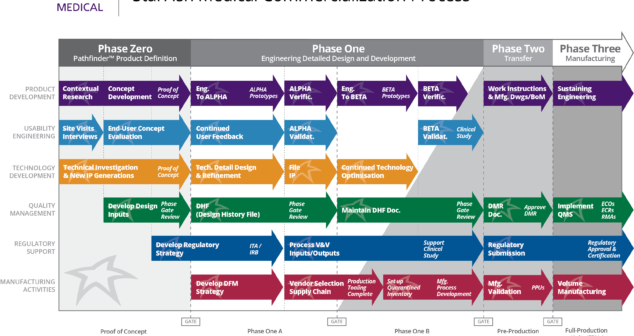
Medical Device Design and Development Process Improvement for Annual Plan: limit improvement efforts, measure them regularly, succeed.
-

The best way to avoid waste is to consider medical device regulatory requirements at a project outset.