Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-
Filter Integrity Biopharmaceutical Manufacturing explores the relationship between microbial retention tests and integrity tests essential for validating the effectiveness of filters in critical applications.
-

Ensuring on-time medical device manufacturing builds are done in a timely manner requires coordinating across several teams including engineering, manufacturing, and quality. Visual Project Management (VPM) and Manufacturing Production Schedules (MPS) help improve manufacturing performance and meet build delivery deadlines.
-

33 tips and examples from commercialization experts for manufacturing a medical device.
-

Medical Device Transfer to Manufacturing tips and examples from building a solid foundation to logistics.
-

Considerations for designing UDI labels for medical devices include ANSI/ISO Parameter Values, label verification and durability.
-

Questions to evaluate your capability post medical device transfer and reinvest in your product to add value.
-

Writing and implementing standard operating procedures (SOPs) for the manufacture of medical devices is required by ISO 13485, FDA, and other regulatory bodies. An SOP is a set of written instructions that documents a routine or repetitive activity that is followed by employees in an organization. The development and use of SOPs are integral parts of a successful quality system. They provide directions to perform a job properly and consistently to achieve predetermined specifications and quality end results. SOPs address all requirements to complete the job or process safely and effectively.
-
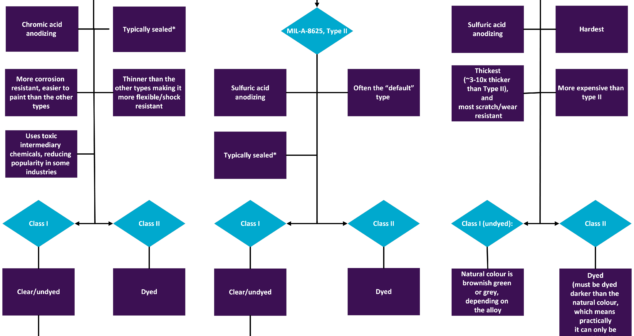
Features and benefits of different Mil-Spec Anodizing Types and Classes of anodized coatings in North America.
-
Medical Device New Product Introduction (NPI) & Commercialization lessons for medical device strategy, design, development, and NPI success.