Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

Medical device color management overview of why colour-rendering accuracy is critical for LEDs CRIs and Full-Colour Imaging.
-

FDA ASCA Pilot Program is expected to bring medical devices to patients and users more efficiently by using ASCA-accredited labs.
-

Biotech in medical device companies: 3 compelling reasons to pursue a Bio Services career in medical devices.
-

COVID-19 friendly brainstorming meetings identifies 5 benefits where virtual brainstorm results outperform in-person brainstorming results.
-
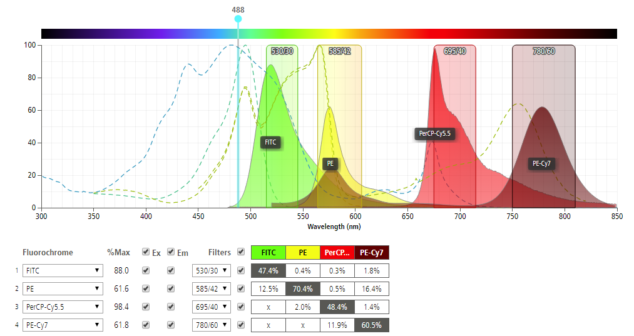
Eight tips to help successfully implement a POC diagnostic platform optical detection with a disposable microfluidic cartridge.