Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

Zombie Microfluidic Cartridge early indicators to determine if a cartridge will function as designed or fail during the assay protocol run.
-

Building an affordable customized Microfluidics Microscope set-up to view droplets as they are being generated.
-
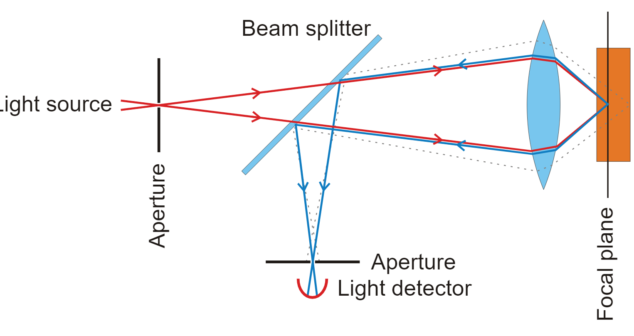
Optical Imaging Biomedical Devices integrate optical imaging with conventional medical imaging devices to enhance biomedical imaging.
-
Commonalities and points of potential confusion of the “big 4” incoherent-light hazard classification and standards for medical devices.
-

Interference Testing, Strategy, Detection Case Study examines POC medical devices assay verification guideline CLSI EP07-A2.
-

How an Autonomous Clinical Chemistry Assay diagnostics design with kinetic read out helps mitigate interference challenges.
-

Considerations for selecting medical device camera image sensors including CCD vs. CMOS and monochromatic vs. color varieties.
-

How to identify hidden costs in medical device Verification & Validation along with mitigation measures to reduce the expense.
-

XYZs of Medical Device Vibration Testing shares the basics of vibration testing from equipment to key concepts and definitions.