Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-
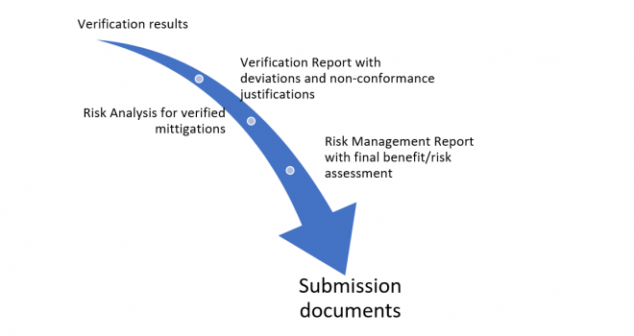
Author describes her experience submitting a medical device application (ventilator) under the Health Canada Interim Order.
-

Knowing which medical device standards apply to your device is an essential part of successfully bring a medical device to market.
-

Health Canada improvements: Getting new devices to market, strengthen monitoring and follow-up and provide more information to Canadians.
-

FDA provides a roadmap to follow for premarket submission in their guidance on Content and Format of Non-Clinical Bench Performance Testing.
-

Overview of Device Establishment Inspection Processes and Standards Update that establishes uniform processes and standards for FDA Inspection Process.
-

9 tips for 510(k) submission along with lessons learned from a recent successful experience submitting a medical device to the FDA.
-

16 things to consider for the Medical Device Regulations (MDR Deadline) and changes you need to make in your Quality Management System.
-

7 interesting outcomes of the 2019 Health Canada Action Plan on Medical Devices initiatives and alignment with international regulators.
-

The Bleeding Edge explains how 510K cleared medical devices are clinically proven to improve patient outcomes and save lives.