Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

Many techniques can be employed in order to simplify formal verification testing. These techniques expedite the product development process and reduce cost….
-
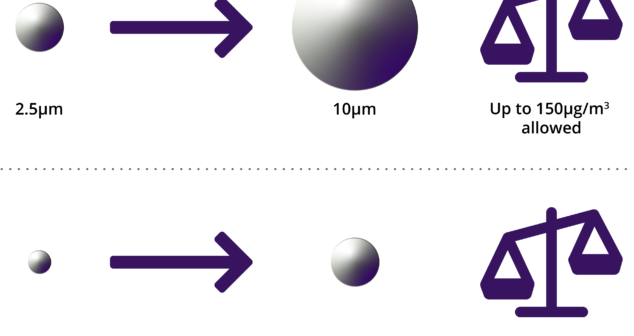
Pre-Testing for ISO 18562-2:2017; how to approach in-house pre-testing and add confidence to 18562:2-2017 formal testing.
-

Outlines how medical device designers can perform in-house Medical Device Drop Test per IEC 60601-1 before third party testing.
-
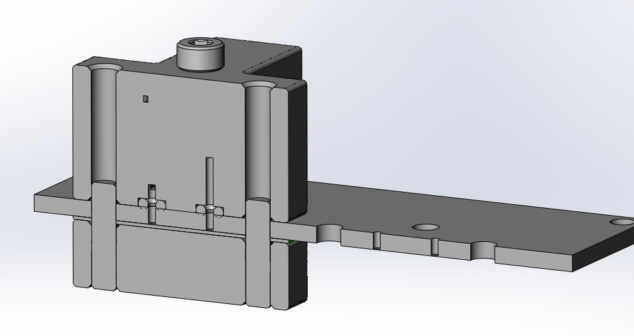
Overview of three physical options for Medical Device Test Fixtures to be used for physical testing fluid connections.
-

Microfluidic Optics explores ways that lenses can be implemented on microfluidic cartridges can play an important role in microfluidic platforms,
-

Optical Safety for Ophthalmic Instruments covers safety limits and maximum exposure limits of wavelengths in the IR, UV and visible light spectrum.
-

Maximize medical device optical signals by keeping sensor and control signals correlated and other strategies are discussed in this article.
-

Microfluidics for molecular and serological diagnostic tests enable shorter turnaround times, high accuracy results, and lower cost per test.
-
Simulated Fluids for POCT Development enable testing using a liquid that mimics the critical properties of the desired clinical sample.