Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-
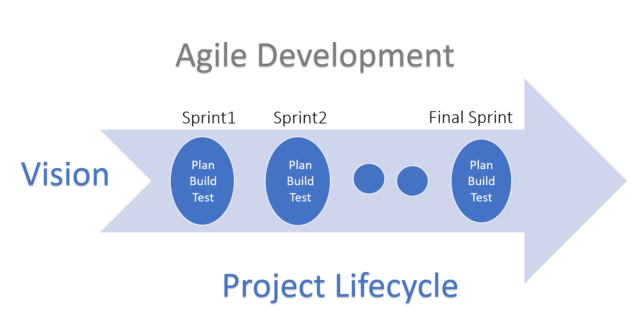
Agile methodology in Medical Devices Product Development methodology is supported by an FDA guidance and IEC standards.
-

Proper documentation is an up-front investment of time and effort that pays big dividends later in medical device development.
-

CPU Modules can be incorporated onto a custom PCB in a medical device or IVD providing a powerful CPU on a product specific PCB without designing the CPU onto their board.
-

Under-estimate the time to construct and debug prototypes? Tips on saving time (money) during the build and debugging of a prototype.
-
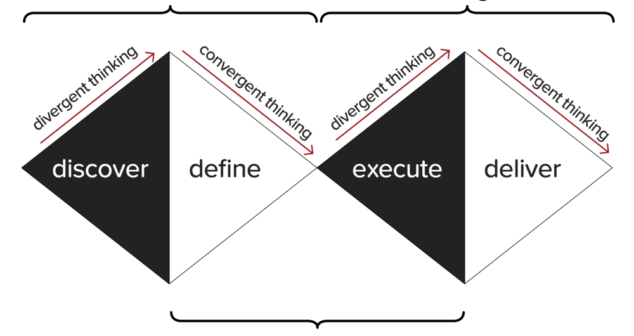
Kepner-Tregoe’s original Decision Matrix overview with tweaks to make it broader and more robust.
-
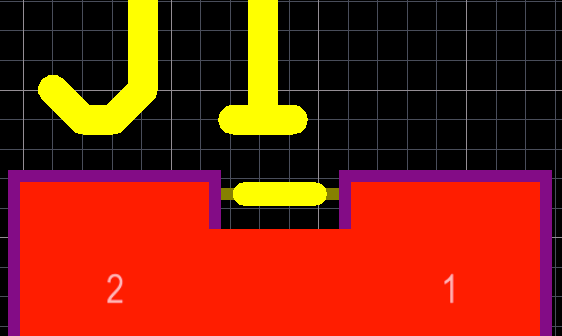
PCB design tips to design your board for ease of assembly, testing, and modification every time you build them.
-

11 medical device development mistakes that surfaced frequently in a variety of products. Avoid them to reduce cost and time to market.
-

How to maximize your biotech R&D budget? Form an academic-industrial partnership. 5 tips for a successful academic-industrial partnership.
-
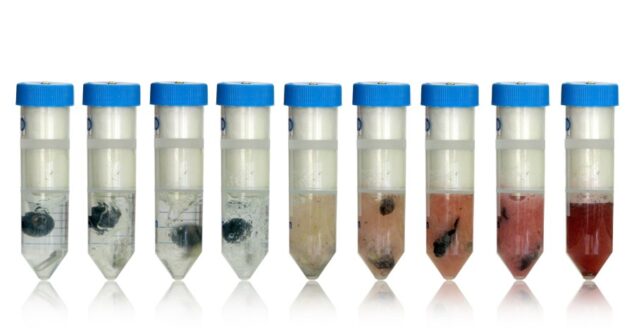
Overview of magnetism and rare earth magnets with a novel medical application homogenizing tissue samples (IE liver or brain) using rare earth magnets.