Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

This article outlines the core elements of the PCCP framework, the types of modifications it applies to, and how the FDA expects manufacturers to use it in practice.
-
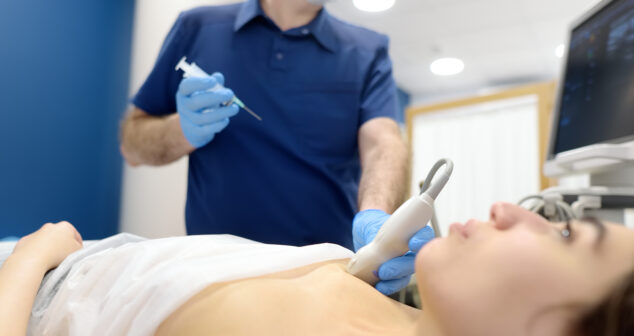
The U.S. Food and Drug Administration (FDA) and the Office for Human Research Protections (OHRP) have released a draft guidance document, Considerations for Including Tissue Biopsies in Clinical Trials, issued in January 2025. It provides recommendations for sponsors, investigators, institutions, and Institutional Review Boards (IRBs) on the safe and ethical incorporation of tissue biopsies in clinical trials.
-
In May 2024, the FDA released an updated guidance document to help manufacturers, third-party service providers, and healthcare facilities determine whether their modifications fall under the category of remanufacturing. This guidance enables these entities to continue ensuring patient safety, regulatory compliance, and the effectiveness of remanufactured medical devices.
-
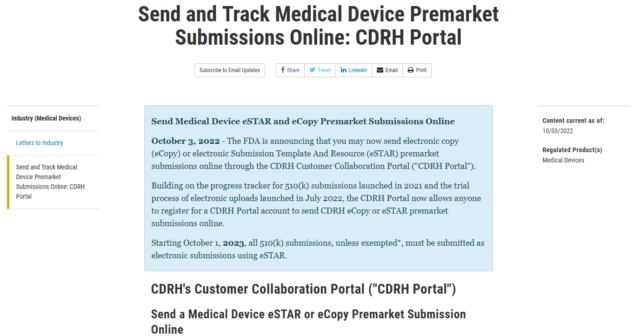
Overview of eSTAR, a joint Health Canada and FDA program streamlining medical device submissions with info on how to apply for the program.
-
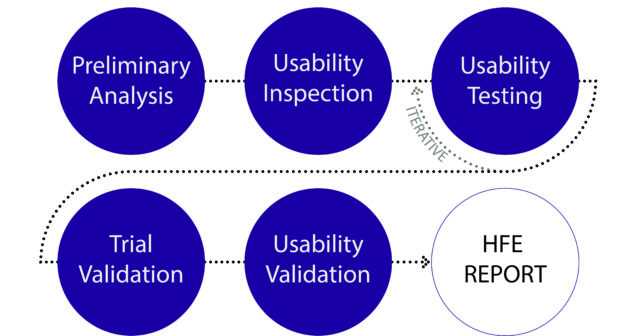
Did you know that this year FDA's Human Factors Premarket Evaluation Team have a priority review hit-list for medical devices?
-

Expedited Access Pathway (EAP) is promising for timely approval and access to devices for life-threatening or irreversibly debilitating conditions.