Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

Explanation of changes in medical device symbols and practical implications as a result of the ISO 15223-1:2021 Symbols Update.
-
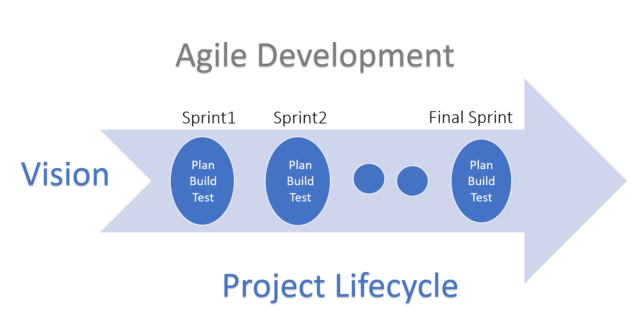
Agile methodology in Medical Devices Product Development methodology is supported by an FDA guidance and IEC standards.
-

Data de-identification should be the first option to consider when dealing with Personal Health Information (PHI) security.
-

Meeting with the FDA can put almost everybody in the company in panic mode. Benefits of FDA meetings and what to expect.
-

Medical device labeling includes all information provided with the device. Article identifies jurisdictions and associated "do's and don'ts".