Design and Development Case Studies
Breakthrough Product Case Studies
We help medtech innovators throughout North America overcome challenging technology obstacles to create breakthrough products that improve health and save lives. The FDA Breakthrough Devices Program provides several advantages for novel devices that meet the inclusion criteria and can significantly reduce a product’s time to market. View a sample of our medical device design and development case studies above.
A Focus on Creating Client Value
“Over the next decade, we want to help 100 of our clients become worth more than $100 million each. It’s not about designing the plastics a little better. It’s not about an incremental cost improvement. Those things are important, but the biggest thing is making sure that what we’re designing is going to have a clinical impact. We should be focused on the long-term value.” – Scott Phillips, StarFish Medical Founder and CEO
Patents
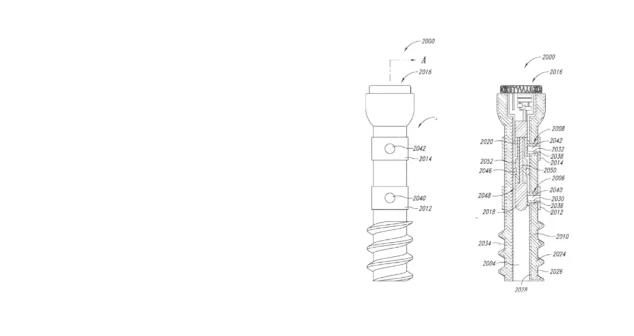
StarFish patents span over 175 Cardiovascular, IVD, Ophthalmology, Optics, and Ultrasound medical devices. Examples include 3D ultrasound from inside the beating heart, spinal surgery for ALS patients, pediatric ventilators, ingestible capsules, and laser platelet analysis for safer transfusions. Several of these are included in our medical device design and development case studies above.
Successful medical device exits for startups often involve a combination of 18 important business factors. One factor is strong intellectual property protection through patents and trademarks to create a barrier to entry for competitors.
Innovation Case Studies
Innovation is central to StarFish culture. We help clients define, design and manufacture innovative devices that optimize founders’ value using our guiding principles of: We look deeper and solve the right problems; We are open and welcome accountability; We get better all the time; and We cut to the chase. Starting with our hiring process, we look for people who embody our guiding principles and demonstrate innovation and team spirit from an early age.
Regulatory Affairs
Navigating the journey from product design to regulatory clearance is no small feat. The path is riddled with obstacles that can delay your time to market and increase risks. To confidently steer through these complexities, you need a partner with deep expertise and a proven track record.
New Product Introduction (NPI)
StarFish Medical employs cutting-edge NPI strategies to deliver products that truly serve patients and clinicians. By performing all medical device product engineering services activities in-house, we ensure a streamlined, lower-risk process when transitioning to full production.
Discover more medical device case studies on our Omnica website.