Wearable medical devices in outer space – Apollo biomedical telemetry
Collecting and transmitting biomedical telemetry has never been easier. You can integrate a few off-the-shelf integrated circuits to collect data like ECG waveforms, pulse oximetry, and temperature. Add a few more off-the-shelf parts, and you can fling that data anywhere in the world across the internet.
It wasn’t so easy in 1962, when President Kennedy announced to the world that the United States was going to the moon by the end of the decade, and the Apollo program was born. Engineers were faced with the problem of collecting vital biomedical data from the astronauts and beaming it back in real-time to mission control so that flight surgeons could monitor the health of the astronauts during the mission as well as collect important scientific data.
Compact wearable biosensors were in their infancy: the now-ubiquitous Holter monitor had just recently been invented. Digitizing and transmitting biomedical data in real-time across vast distances was mostly in the realm of fantasy.
NASA rose to the challenge and produced an effective bioinstrumentation system for Apollo, an achievement which is particularly impressive considering the technological limitations present at the time.
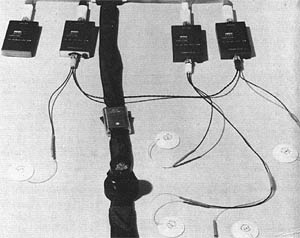
Apollo astronauts wore an instrumentation rig designed and manufactured principally by NASA contractor Spacelab. It consisted of an ECG, impedance pneumograph, and an oral temperature probe. The instrumentation was an evolution of the technology used in the earlier Mercury and Gemini programs. In particular, the oral temperature probe was a major improvement in astronaut comfort over the core temperature probe used in Mercury. This rig was mounted in a “bio-belt” that the astronauts wore inside their spacesuits (see image at top of this article).
The impedance pneumograph in particular is a milestone technical achievement. Essentially, a small AC current is applied to the chest cavity to measure the impedance between two electrodes. As the subject breathes in and out, changes in impedance are used to quantify both breathing rate and depth. Impedance pneumography is a widespread technique today but was essentially unheard of in the early 1960s. For the Apollo program, NASA and Spacelab took the concept straight from university labs, miniaturized it, and deployed it during actual spaceflights.
While the sensors themselves were impressive for the 1960s, equally impressive was the system used to take these biosensor readings and transfer them back to mission control in real time.
The Command Module and Lunar Module both carried eight-bit analog-to-digital converters weighing over 20 pounds each. Amplified signals from the biosensors were multiplexed into these converters along with analog signals from other spacecraft systems and sensors. The digitized signals were inserted into a pulse-code modulated (PCM) bitstream which formed the primarly telemetry feed. This bitstream was then phase modulated onto a 1.024 MHz subcarrier and multiplexed with other communications channels (such as voice, TV, and a ranging signal). The baseband signal was then further phase-modulated onto an S-band microwave carrier and beamed back to the ground stations on earth, where the process was reversed.
This digital system was used when the astronauts were inside the spacecraft. However, the astronauts also needed biomedical telemetry during extravehicular activities (EVA). The biosensors were detached from the spacecraft’s primary telemetry system during moonwalks, and it was not practical to put a 20-pound digital telemetry unit in a spacesuit. Instead, the biomedical signals were transmitted as frequency-modulated subcarriers on the AM VHF link from the astronaut’s suit to the Lunar Module. The frequency-modulated signal was recovered by the electronics aboard the LM and mixed into the baseband for the S-band link back to earth.
The system worked very well. For the most part, biomedical data collected during the Apollo program was used to further our understanding of the human body’s reaction to the space environment.
In one instance, telemetry from an astronaut’s suit was used to diagnose a heart condition during a spaceflight. During the return of Apollo 15 from the moon, lunar module pilot James Irwin developed bigeminy on his ECG (shown below). This was cause for consternation on the ground. Flight surgeon Charles Berry stated that had Irwin been on earth, he would have immediately been transferred to the ICU for treatment for a heart attack. Ultimately, since Irwin was in an ICU-like 100% oxygen environment inside the spacecraft, he was instructed to simply rest and recover. Irwin landed safely, although his heart problems continued for the rest of his life.
Peter Kazakoff wrote this blog as a StarFish Medical Electrical Engineer. A graduate from the University of Victoria, Peter received the Chris Denny Memorial Award for Innovation in 2017. He worked on a variety of medical device projects at StarFish, and recently relocated to Quebec to follow his dream and join the Canadian Space Agency Engineering training program. We wish him great success!
Image: NASA / Public Domain
Learn key success factors in designing, developing wearable medical devices plus the importance of selecting appropriate adhesives.