Designing Point of Care Diagnostics
It is all about the sample
Point-Of-Care Testing (POCT) has seen a large uptake over the past year driven by the need for on-site testing due to the COVID-19 pandemic. We see that often companies focus their attention on one specific sample type and subsequent sample preparation (e.g. blood or nasopharyngeal samples) and build a platform and market around the constraints of that sample type.
Although this strategy has proven successful, it would be truly revolutionary to have platforms breach through these constraints by being able to process a multitude of different patient specimen. This review outlines some of the considerations that go into designing Point of Care diagnostics systems.
Sample preparation for molecular diagnostics
Serological POCT (based on antibody-binding) often delivers a fast result with a relatively easy workflow but the tests suffer from lower reliability. In terms of analytical performance, only molecular-based systems (e.g. systems that make use of processes like Polymerase Chain Reaction (PCR) or isothermal amplification) have the potential to truly displace the need for laboratory-based testing. A molecular test can generally be divided up in the following steps:
- Sample acquisition
- Sample preparation
- Amplification of genetic material
- Signal detection.
Generally, steps 3 and 4 are relatively straightforward if a clean sample is readily available and many on-market tests use similar strategies for amplification and signal detection. In contrast, sample preparation can be challenging. Most of the innovation and differentiation between test platforms centers around the strategy for sample acquisition and preparation.
Which specimen should you collect?
Not every specimen is suitable for detection of every pathogen or disease. For example, different specimens from a COVID-19 patient carry different loads of coronavirus and some specimens are unsuitable for detection of COVID-19 at all. At the onset of the pandemic, researchers tried to determine the best sample type for detection of SARS-CoV-2. They found that sputum (distinct from saliva) is the best for detecting coronavirus however, sputum is very difficult to collect. After sputum, a nasopharyngeal swab has the second greatest chance of collecting coronavirus. That is why the vast majority of COVID-tests use nasopharyngeal samples for analysis. Other specimens that are used for detection of COVID-19 include nasal samples (by sampling mid-turbinate as opposed to deep inside the nasal cavity), oropharyngeal samples and saliva.
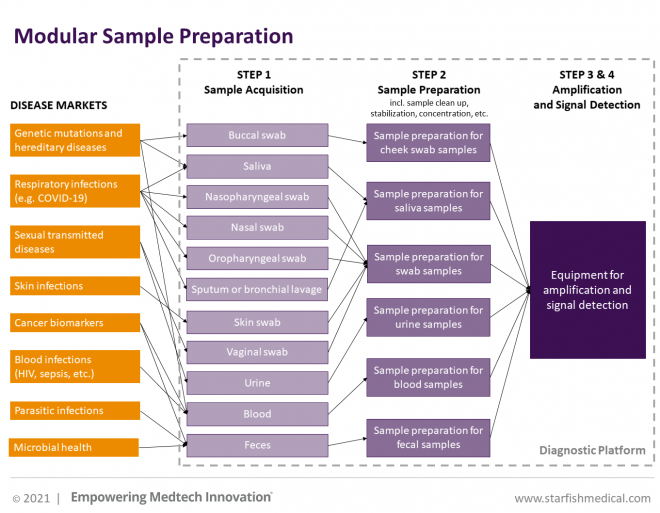
Nasopharyngeal samples are often used for detection of respiratory infections. For other applications like Sexually Transmitted Infections (STI’s), the specimens that can be used are urine or genital swabs whereas Human Immunodeficiency Virus (HIV), sepsis and cancer biomarkers can be detected in blood. Fecal matter could be used for detection of parasitic infections or for screening of microbial health. Buccal samples (cheek swab) or saliva are often used for screening of genetic mutations or hereditary disease profiling. This list is not comprehensive but it outlines how the compatibility of a sample type with a platform determines what diseases can be targeted by that platform and what markets can be addressed. The more specimens that are compatible with a platform, the more markets that can be addressed and the more value is associated with the diagnostic platform.
Who is taking the sample?
Not every specimen is easy to collect. For example, it is not easy to use a nasopharyngeal swab on yourself and reach deep enough into the nasal cavity. Feces and sputum are also hard to collect. It is easy to collect a drop of blood, but a larger volume requires a trained professional. Choosing the right specimen for POCT is therefore a balance between ease-of-use and assay compatibility to drive analytical performance. The choice of specimen collection can also inadvertently exclude some patients from being able to use the test. For example, it is almost impossible to collect a saliva sample from a patient that is intubated.
Where is a specimen stored and how long is it stable for?
After collection, the genetic material needs to be released and protected from enzymes that chew up DNA and RNA. Many of the solutions that are used for storage are not compatible with direct amplification. These solutions can also dilute the genetic material and decrease the assay sensitivity. For example, the Virus Transport Media used for storage of nasopharyngeal COVID-19 swabs inhibits polymerase enzymes required for amplification. It also dilutes the virus. That is why the sample often needs further processing prior to analysis. A detailed profile of sample stability and all of the intermediate sample states needs to be demonstrated as part of a regulatory submission.
What do you have to do with the specimen before amplification?
Especially in POTC, it is important that the sample preparation is as simple as possible. Direct loading of the sample into the system without any preparation is the easiest approach, but may not be possible. Many specimens contain inherent inhibitors of enzymes and other components required for amplification. Samples can also interfere with the signal read out (e.g. autofluorescence). It is often necessary to clean up the sample and remove inhibitors from the specimen. Feces, blood, urine and saliva are notoriously difficult to analyze because of their inherent inhibitory properties. Overcoming inhibition is easier for samples that are more similar from patient to patient (e.g. blood and urine) as compared to samples that can be very different from patient to patient (e.g. saliva and feces). The other sample preparation that needs to be considered is concentration of the sample. This will enhance sensitivity of the assay and may be required to meet analytical performance targets.
Modular approach
Solutions that allow specimens to be analyzed on a platform can be integrated in the product at different system levels. For example, overcoming inherent sample inhibitors can be achieved by sample preparation or by using specially engineered enzymes for amplification that are more robust and immune to inhibition. More specific product solutions like special enzymes are generally restricted to specific sample types and do not allow bridging into other specimens. Each sample type would need their own unique solutions. A platform that uses a modular approach could be the solution. In this setup, the sample preparation would be separately and uniquely designed for each sample type while the platform remains the same regardless of what specimen is used. This type of platform can be slowly expanded by onboarding different specimens depending on business/market needs. The platforms’ ability to multiplex (to be capable of running entire disease panels) and affordability of consumables are also critical considerations that need to be designed into the system from the start.
Conclusion
Because of the COVID-19 pandemic, many platforms have been released with solutions for nasopharyngeal, nasal, oropharyngeal and saliva as compatible sample matrices. It is exciting to see what solutions are being developed to branch into other sample types like fecal matter, urine and blood. Companies designing Point of Care Diagnostics that make their platform truly universal and agnostic to the sample matrix will certainly have a huge advantage in the POCT market.
Joris van der Heijden is Bio Services Program Manager at StarFish Medical. Joris received his PhD in infectious diseases from UBC. Prior to starting his work at StarFish, he was the Director of R&D at Spartan Bioscience where he lead the development of a point-of-care COVID-19 diagnostic test.
Join over 6000 medical device professionals who receive our engineering, regulatory and commercialization insights and tips every month.
