
Using Medical Device Symbols – ISO 15223-1:2021
ISO 15223-1:2021 Key updates
To support consistent communication, it’s important to have international standards that standardize the look and definition of symbols used in medical devices. ISO 15223-1 is one of those key standards. In July of 2021 the International Organization for Standardization (ISO) released ISO 15223-1:2021 Medical devices — Symbols to be used with information to be supplied by the manufacturer — Part 1: General requirements. This is a fourth edition update from the third edition released in 2016. Its purpose is to specify what symbols can be used to express information about a medical device. It includes the symbols themselves and the meaning of each symbol.
Symbols are a universal language. When looking at smiley and sad faced emojis anyone from around the world can differentiate the meaning between the two. That is the power of symbols.
In the right context symbols can convey a significant amount of information in a small amount of printed or digital space. That is why they are valued and encouraged for use as part of medical device labelling. In September 2016, the FDA began permitting symbols without the need for adjacent explanatory text as long as a glossary is provided in accompanying documentation.
The most interesting change in the fourth update is the addition of 25 symbols, many of which allow better alignment with the EU’s Medical Device Regulations (MDR) 2017/745. Here is a summary of the types of new symbols with an example of each:
- Addition of Importer and Distributor symbols

- Five new symbols related to sterilization and sterile barrier systems
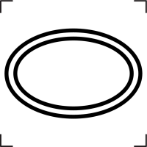
- Six symbols that convey the addition of noteworthy substances including one for hazardous substances

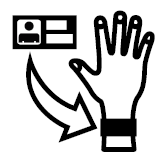
- Five symbols for management of patient data such as a symbol for patient name and date medical information was entered as a record
- Seven general symbols covering items like repackaging, UDIs (Unique Device Identifier), single patient multiple use and country of manufacture to name a few

Additionally, the title of the standard changed from “…medical device labels, labelling and information to be supplied…” to “…information to be supplied by the manufacturer…”.
The phrase, “information supplied by the manufacturer”, expands the definition of labelling to include physical labels and other information such as technical descriptions and instructions for use. This is nicely depicted in Figure 1 of ISO 15223-1. As a result, the previous definition for labelling has now been removed.
For more details on the specific changes in ISO 15223-1:2021, including the new symbols and their use, purchase a copy of this standard from a reputable source such as the ISO webstore.
Practical Implications:
As of late August 2021, the FDA’s Recognized Consensus Standards database and the EU’s Harmonized Standards list only recognize the 2016 edition of ISO 15223-1. It is expected that the FDA and the EU will update their lists to include the new edition. In the meantime, there is no reason to delay the use of this standard as it is published by ISO and the new symbols have been validated using ISO 15223-2, Medical devices — Symbols to be used with medical device labels, labelling, and information to be supplied — Part 2: Symbol development, selection and validation.
Devices already in the field do not require updates to their labelling. Manufacturers of devices in production should examine this standard for its applicability. Though it is encouraged to use these symbols, they are not fully mandatory in all countries unless specifically required to meet regulations and standards that require compliance.
Examples of this include the statement from the EU’s MDR 2017/745, Annex I, Chapter III – 23.1 (h), which states, “Where appropriate, the information supplied by the manufacturer shall take the form of internationally recognized symbols.” As well, IEC 60601-1:2020 Medical electrical equipment – Part 1: General requirements for basic safety and essential performance, Section 7 calls out the use of several symbols as described in ISO 15223-1. It is important to note that IEC 60601-1:2012 only refers to the 2016 version of ISO 15223-1.
But overall, my general take home message is that a re-examination of labeling regulations within jurisdictions of interest to you is a relevant activity at this time.
It is also important to note that ISO 15223-1:2021 is not the only international standard on the use of symbols in medical devices. Others include ISO 7010:2019 Graphical symbols — Safety colours and safety signs — Registered safety signs and IEC TR 60878:2015, Graphical symbols for electrical equipment in medical practice. As well, the origin of the symbols themselves come from sources such as the symbols catalogue of ISO 7000 and IEC 60417.
Lastly, ISO 20417:2021 Medical devices — Information to be supplied by the manufacturer was also released this year. It is a brand new standard that outlines requirements for all kinds of information supplied by medical device manufacturers from labels on devices to the accompanying documentation such as user guides and service manuals. This one is also a worthy read.
Conclusion
The updates within ISO 15223-1:2021 align with both the still youthful EU MDR and the movement towards a universal language of symbols. A picture may be worth a thousand words, but a well-defined and used symbol is priceless for its ability to convey accurate information to medical device users especially for complex Enterprise Partnership projects.
Title Image: © 14101140 – Can Stock Photo / everythingpossible
The symbols taken from ISO 15223-1:2021, Medical devices – Symbols to be used with information to be supplied by the manufacturer – Part 1: General requirements, is reproduced with the permission of the International Organization for Standardization, ISO. These symbols can be obtained from any ISO member and from the webstore of the ISO Central Secretariat at the following address: www.iso.org. Copyright remains with ISO.
Deborah Pinchev is StarFish Medical’s Toronto QA/RA Manager. She is inspired to write blogs by her interest in helping companies develop and bring to market new and innovative devices.
