Curious about the checklist, video, and free consultation offers for your medical device concept that appear on our site? Here’s how an entrepreneurial company that is not a StarFish client uses our medical device process and materials to improve their business and medical device success.
Medinovel, a medical innovation start-up, is using free tools from StarFish Medical’s website to help them develop Quickie-Prep– an easy and fast skin prep applicator for the operating room—and make a smoother transition from consumer to medical device products.
“For new medical product developers, Starfish’s tools provides a sanity check as we move through the product development process”, explains Ola Kowalewski, CEO and Co-founder of Medinovel. “It’s really great to have a company like StarFish as a virtual neighbor, with a lot of project experience to draw upon. Their product development framework helps early stage start-ups like Medinovel understand what it takes to bring a medical product to market compared to a consumer product “.
Quickie-Prep turns a conventional multi—step process into a single step with the aim of reducing the likelihood of surgical site infection and improving ease-of-use. By eliminating steps, Quickie-Prep aims to diminish the incidence of cross contamination and provide a more tactile experience for the OR personnel to get their job done.
Medinovel co-founder, Dr. Richard Kowalewski- a world renowned cardiac anaesthetist – had the concept in mind for over 15 years. Regulatory recommended practices have changed in recent years, making it possible to bring Quickie-Prep to life. Prior recommended practice was to use Iodine with three applications. Now recommendations include a chemical which only requires one application.
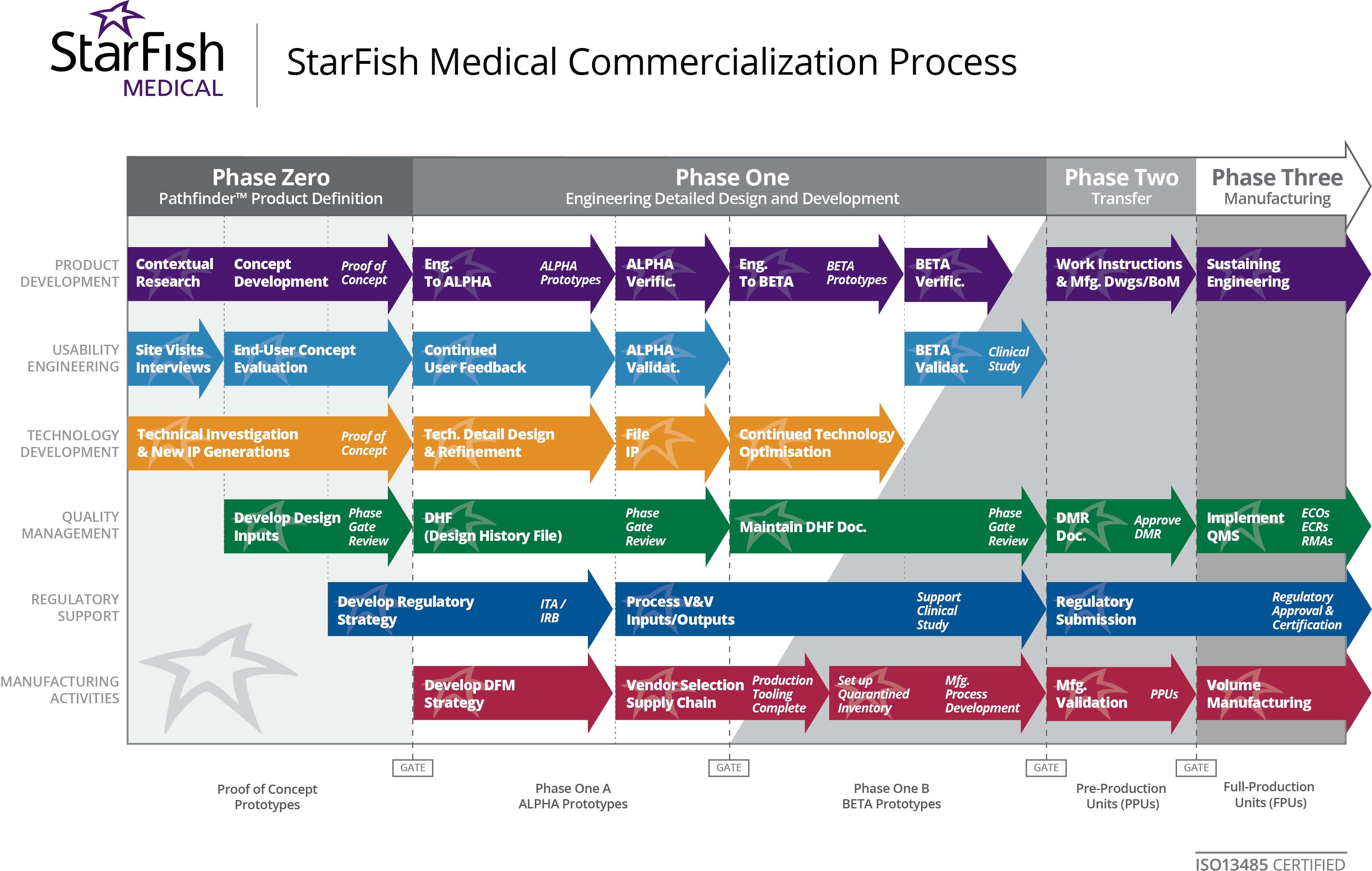
Medinovel and Quickie-Prep are a pivot for Ola’s furniture design start-up. After receiving positive feedback from physicians on an R&D version of her father’s concept, Kowalewski started getting serious about Quickie-Prep. Ola developed a team with government funding that placed in the Top 10 of the 2013 BC New Venture Competition. “BC New Venture pushed things forward. It helped develop and validate our business strategy, and provided important mentorship and advisors within the community.”
Ola explains some of the differences between the original company and a medical device start-up: “The StarFish Development Process Chart is helpful when defining roles and responsibilities, hiring people, and keeping product development in check. I organize responsibilities based on the StarFish process of six main parallel sections. Everything overlaps as a big system working together. As a result, the product is being developed in a holistic manner.
When we filed our IP it was good to check how we were doing in all the other streams, and to consider whether we weren’t forgetting any other important steps. “
And the future? “In the fall we’re looking for funding for our validation phase with clinical trials. We believe Quickie-Prep will be the new skin prep gold standard worldwide. Skin prep is performed on every surgery in the world – so it’s a huge market, growing and not going away.”
I love Ola’s entrepreneurial spirit and am glad she is gaining value from our process and materials. Do I hope she’ll become a client when ready? Absolutely! We enjoy working on cool concepts with motivated leaders. I also want to hear from other readers on how they are using our process and tools.
Scott Phillips is Founder & CEO at StarFish Medical. Scott holds a degree in Engineering Physics from the University of British Columbia. Under his leadership StarFish has grown into a diverse professional organization with clients around the world and 100% focus on medical devices.
Image: StarFish Medical
