This blog covers the optical safety limits and maximum exposure limits of wavelengths in the IR, UV and visible light spectrum for ophthalmic instruments. Optical safety for ophthalmic instruments focuses on ocular exposure limits because the eyes are the most light-sensitive part of the body and ophthalmic instruments are intended to interface directly with the eyes during diagnosis and treatments.
Optical radiation used in medical devices can be generated by a multitude of sources including lamps, LEDs and lasers of widely differing wavelength, duration, coherence and output power. Depending on the wavelength and tissue type, optical radiation is absorbed to varying degrees by the human body. This absorption may result in a number of hazards to the body that ranges from mild irritation to permanent damage, blindness and even death, depending on the wavelength and optical power.
It is extremely important to know the type of optical radiation you are working with and its potential interactions with the human body. How can you protect yourself? What is the maximum allowable exposure levels? If the optical radiation is part of a treatment, is it safe to use?

Optical radiation of differing wavelengths interacts with the human body in different ways. Below are a small sample of these interactions:
- IR: Thermal
- NIR and Red: Photobiomodulation
- UV: Photochemical
Ophthalmic Instruments
Ophthalmic instruments are divided into two distinct groups to distinguish between hazardous and non-hazardous radiation exposure levels as described in ISO 15004-2:2007 “Ophthalmic instruments — Fundamental requirements and test methods — Part 2: Light hazard protection”.
Group 1 ophthalmic instruments do not pose a light exposure hazard. Group 2 ophthalmic instruments pose a potential light exposure hazard. If your instrument could pose a light exposure hazard (Group 2), it is recommended to attenuate all wavelengths as much as possible without reducing the efficacy of the instrument as identified in the “intended use”.
Corneal and Lenticular or Retinal Irradiance:
Group 1
Group 1 Ophthalmic instruments must meet any or all of the following criteria:
- An International Standard exists for the instrument type but no light hazard requirements are included in that standard.
- Individual components of the instrument prevent emissions that exceed the limits for Group 1 instruments. Certification of efficacy must exist for these components. Sources may ensure safety by blocking (filtering) out some of the wavelength components that otherwise would be emitted by the instrument: the spectrum of the unblocked wavelengths must be assessed and verified against the relevant exposure limits.
- Emissions do not exceed the limits specified for instruments in Group 1 as specified in ISO 15004-2:2007 “Ophthalmic instruments — Fundamental requirements and test methods — Part 2: Light hazard protection”
- If the sources of radiation are produced by lasers, only Class 1 lasers (under IEC 60825-1:2001) are permitted for Group 1 instruments.
Group 1 Exposure Limits:
The exposure limits are based on an expected exposure time of 2 hours. The exposure limits are decreased by a factor of 2 for operation microscopes and endoilluminators, and decreased by one-half of the exposure time for instruments designed for continuous exposure. For example, if the total exposure time were 6 hours, then the maximum permissible exposure is reduced by a factor of 3. The exposure limits are listed separately for continuous-wave versus pulsed optical radiation.
The exposure limits have been divided into wavelength bands that produce specific hazards. These hazards are as follows:
- Photochemical damage to the retina due to aphakic light (Blue light)
- Thermal damage to the retina due to visible and infrared radiation
- Thermal damage to the cornea and lens tissue due to near-infrared and infrared radiation
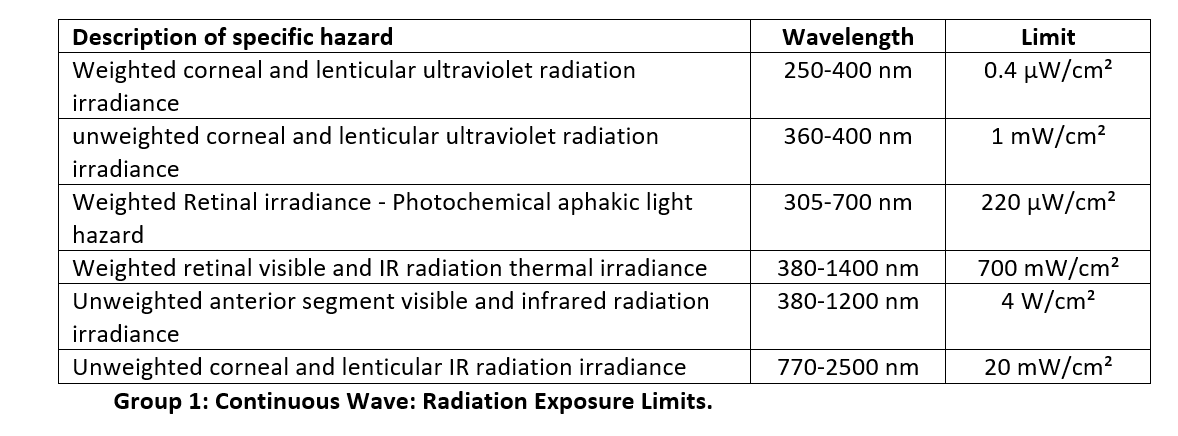
Group 1 Pulsed Optical Radiation Limits:
The pulsed optical radiation limits are based on the continuous wave radiation limits and the time-averaged values of repeated pulses for a given duration. The time-averaged values are calculated by multiplying the number of pulses in a given time period by the maximum energy per pulse over the given time period.
Any pulsed optical radiation source may be considered a continuous wave source if the pulse duration is greater than 20 seconds.
More in-depth calculations of the pulsed optical radiation limits are beyond the scope of this blog, but these limits may be found in ISO 15004-2:2007 “Ophthalmic instruments — Fundamental requirements and test methods — Part 2: Light hazard protection”.
Group 2
Group 2 Ophthalmic instruments requirements:
- Emissions do not exceed the limits specified for instruments in Group 2.
- Any controls to modify the brightness shall indicate fractions of the maximum intensity up to the maximum intensity.
- The appropriate information and cautionary statements shall be provided with the instrument. Refer to Clause 7 in ISO 15004-2:2007 “Ophthalmic instruments — Fundamental requirements and test methods — Part 2: Light hazard protection” for details.
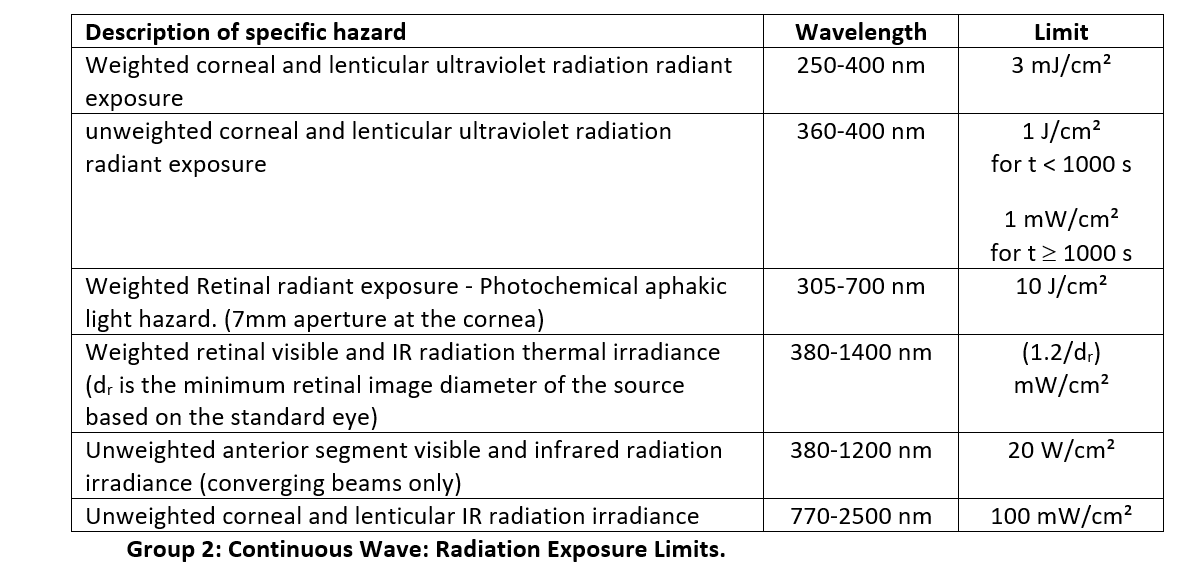
The Group 2 exposure limits are much greater than the limits for group 1: at these higher powers, the sources could pose a hazard to the patient. The measurement requirements are slightly different for assessing Group 2 instruments: consult ISO 15004-2:2007 “Ophthalmic instruments — Fundamental requirements and test methods — Part 2: Light hazard protection” for measurement details.
Group 2 Pulsed Optical Radiation Limits:
Group 2 pulsed optical radiation limits are calculated similarly to Group 1 pulsed optical limits, but using the Group 2 continuous wave limits.
Conclusion
Although ophthalmic instruments are deemed safe because they comply with the ISO 15004-2:2007 standard that limits optical radiation exposure, it is important to minimize the time exposed to Group 2 ophthalmic instrument radiation because overexposure could pose a potential light exposure hazard. Group 1 instruments are much safer. The maximum exposure to Group 1 ophthalmic instrument radiation is 2 hours.
Joshua Hayes is a Senior Electrical Engineer at StarFish Medical. He is skilled in analogue and digital electronics design and PCB layout and has extensive experience designing firmware for embedded systems. A graduate of Camosun College and the University of Victoria.