Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

Ariana and Mark walk through what separates a clinical prototype from a proof-of-concept build, what determines how much testing and documentation you actually need, and where the regulatory line between significant risk and non-significant risk falls.
-

In this episode of Bio Break, Nick walks through both patent types after receiving two of his own in the mail, one of each, from the USPTO.
-

Scott Phillips sits down with Mickey Urdea to examine what actually distinguishes companies that reach commercial outcomes from those that do not.
-

Nigel Syrotuck breaks down the realistic medical device V&V cost and schedule for terminally sterilized devices, picking up after design freeze and walking through each major phase of the process.
-

Nigel Syrotuck examines the hidden medical device sterilization costs that many development teams overlook when estimating the true cost of producing a terminally sterilized device.
-

Nick Allan and Nigel Syrotuck explain exactly how aminoglycoside antibiotics work and why they're so effective at killing bacteria.
-

Graphical mind maps created in online whiteboards offer a low-barrier, highly collaborative approach to early risk analysis in medical device development.
-

Early phase concept development is a weird part of a project lifecycle. It is often the most exciting phase, because the team is exploring possibilities, generating new ideas, and turning a fuzzy opportunity into something real.
-
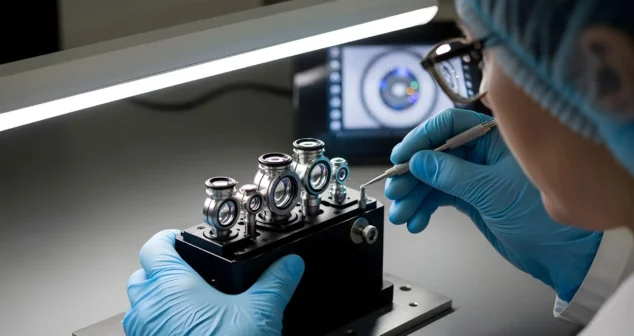
Clinical prototypes must not only function as intended, but also be manufactured, documented, and supported in a way that satisfies regulatory expectations and clinical realities.