
Tools and tips for holding COVID-19 friendly brainstorming meetings
Brainstorm Better with COVID-19 friendly brainstorming meetings
Just because your team is working from home due to COVID-19, doesn’t mean your ability to work collaboratively has to suffer.
Our design team has been working collaboratively between two offices on opposite ends of Canada for more than three years now. Holding a meeting online might not seem as effective as holding one in person, but being able to have all participants at a computer actually offers a number of benefits.
Benefits:
- Less physical limitations: Not being tied down to a physical whiteboard in a physical meeting room allows users to pre-populate the virtual whiteboard with some initial “seed” ideas to get the ball rolling, before the meeting takes place
- Search and share references: Diagrams, images, and even videos can be brought up and posted to the virtual board.
- Sketch digitally: No longer are you bound to using smelly, dried up white board markers. You can now draw with lines and shapes over photographs. If you have a tablet or cell phone, you can use a stylus or draw with your finger for more accuracy.
- Level the playing field: It can sometimes be challenging to have some participants in a board room while a few off-site participants join over conference call. These off-site participants can often be KOLs or subject matter experts. Having everyone attend through video conference puts people on even footing to participate.
- Access Whiteboard contents any time: There’s no longer a need to take a photo of your whiteboard before it gets erased. If anyone has follow up thoughts or ideas, they can be shared on the virtual white board following the meeting.
Use these tools and tips to easily lead fun and effective COVID-19 friendly brainstorming meetings.
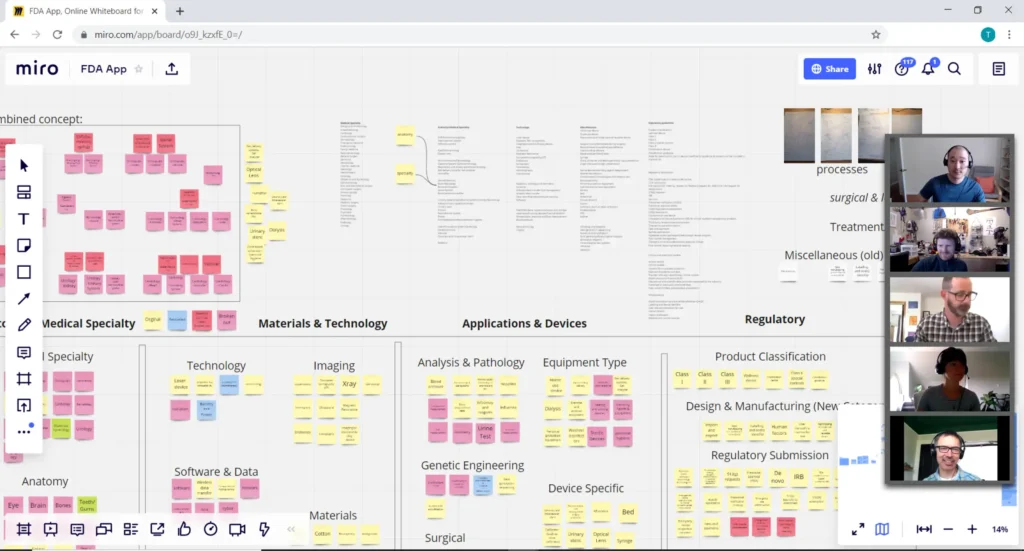
Tools:
Virtual whiteboard (obviously): A virtual collaborative white boarding tool is critical. Many web-based applications allow multiple users to collaborate simultaneously. Our personal favorite is Miro.
Video conference tool: Miro happens to have a video conferencing feature built in. But if the white boarding tool you’re using doesn’t have one, a service like zoom works well. Video conferencing may be preferred anyway in case somebody needs to share their screen.
Facilitator: The person who sets up and leads the meeting. As the facilitator, you are responsible for keeping everyone focused on the topic.
Participant: As a participant, you are responsible for attending the meeting and doing any reading assigned to you beforehand.
Pre-meeting activities:
- Do your homework: Brainstorming meetings are much more fruitful when the facilitator and a few key participants have prepared for the brainstorming session ahead of time. Finding reference material and completing some initial brainstorming beforehand provides a springboard from which to start generating even better ideas.
- Get everyone up to speed.
- Make sure that participants are familiar with the project and its challenges so that time isn’t lost briefing people about the project during the meeting.
- Nail down the exact problem or problems you’re looking to solve. It helps to have a well-defined problem statement (or statements) to pose to the group.
During the meeting:
- Encourage everyone to keep their webcams on: Being able to use hand gestures or read people reactions can be a big help.
- Get visual: Luckily tools like Miro offer lots of features to help sketch, build mind maps, diagrams, and post images and links to competitive products, promising technologies etc.
- Don’t critique or debate ideas: Go for quantity and encourage wild ideas, silly ideas (within reason) can keep the session fun and engaging.
When done with the right tools and techniques, virtual COVID-19 friendly brainstorming meetings can be a fun and effective way to conduct sessions.
Have a look at our Brainstorm Better PDF for more tips on improving both quantity and quality of your brainstorming ideas.
Tim Park is a StarFish Medical Industrial Designer based in our Toronto office. His experience includes consumer and medical device products. Clutter is not in his vocabulary.
Read how StarFish Medical led a consortium that created a Ventilator 2.0 therapy device in record time.
