Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-
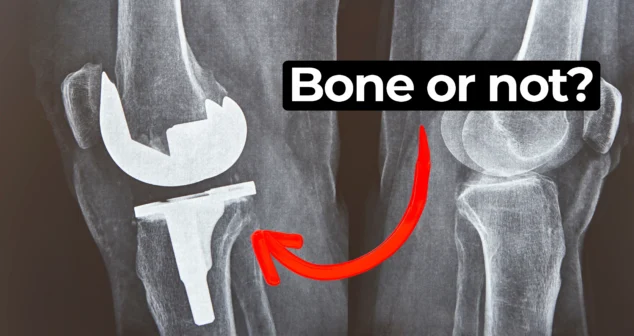
In this episode of MedDevice by Design, Ariana and Mark dive into the biomechanics and materials science behind osseointegration for implants.
-
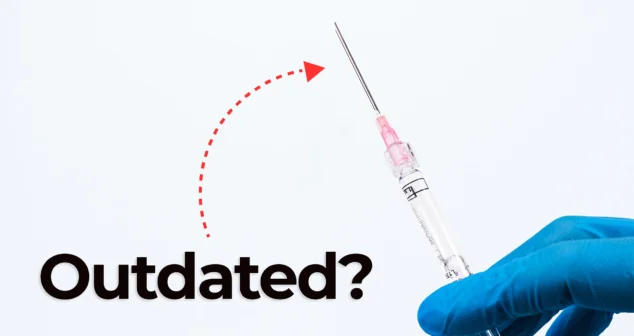
Nick and Nigel dive into the world of jet injector drug delivery. This needle-free method, made popular in science fiction and real-world vaccines, is still used today.
-

Ariana and Mark explore how accommodative intraocular lens technology may one day restore natural vision for people who require cataract surgery or suffer from presbyopia. As Mark shares, traditional bifocals are not ideal, and new lens solutions may offer better outcomes.
-

Many clients now request their devices to look and feel like Apple products. But achieving that level of simplicity and elegance is not as easy as it seems.
-
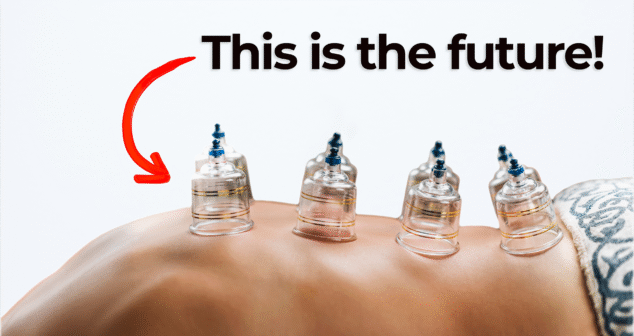
Nick and Nigel explore a surprising approach to injection pain reduction using suction technology. What started as an unusual product order at StarFish Medical led to important insights on improving patient comfort during injections.
-

You have a great MedTech innovation idea and are trying to decide whether to build a team to commercialize a medical device…
-

We explore the world of brain-computer interfaces (BCIs) and the challenges of capturing thought into action. Mark Drlik and Ariana Wilson walk through how these systems translate brain activity into control signals for devices—without needing surgical implants.
-

Nigel and Nick explore microneedle drug delivery—a growing field in medtech that aims to improve patient comfort and treatment compliance. While the term "microneedles" may sound futuristic, this technology has been around for years.
-

The impact of shear stress is critical to effectively design medical devices that handle biological fluids such as proteins or cell culture media. For example, non-physiological shear stress (NPSS) on blood is a key factor because hemolysis (cell rupture) could occur due to accumulated stress.