Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

Entrepreneurial company uses StarFish Medical device process and materials to improve their business and medical device success.
-

Engineering expertise in design and development lessons learned in the years since school – both in and out of the medical device industry.
-

Six ways to manage and communicate medical device project relationships without losing the essence of the development innovation or wasting money.
-
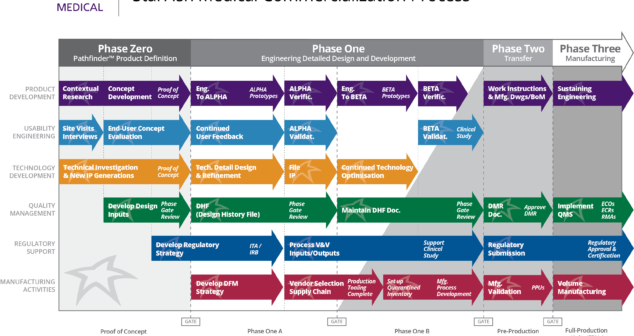
Medical Device Design and Development Process Improvement for Annual Plan: limit improvement efforts, measure them regularly, succeed.
-

Medical device margin matters: In initial medical device product definition always consider the margin in device development discussions.