Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

Four areas that can make or break a new medical device development project from the start. Experts identify actions and offer advice that helps ensure new medical device projects start on the right foot.
-

Engineers, regulatory, manufacturing and optics experts share their experiences and lessons learned commercializing hundreds of medical devices with optics components and interacting with optics engineers.
-

Three common elements can be observed in biomedical product development projects that are doomed to failure. They are the Three “P’s” – Platform, Point of Care, and Program Manager.
-
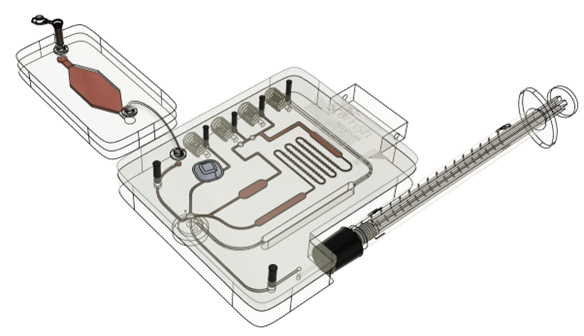
How a ready-made modular proof of concept system for rapid prototyping Point of Care Diagnostics can expedite and de-risk product development.
-

Overview of the Canadian SR&ED program and a 5 step high-level guide on how to prepare an SR&ED tax form (or T661).
-
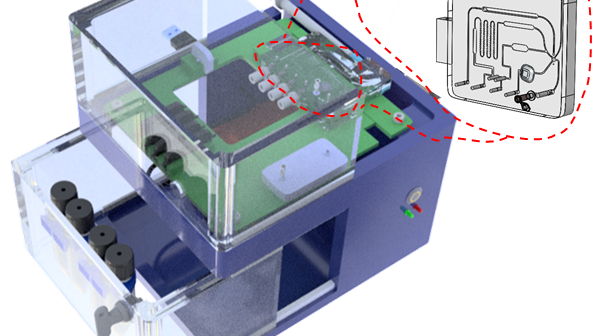
The modular platform for transferring immunoassays acts as an intermediate bridge and is a significant step towards streamlining the product development process for diagnostics.
-

Medical Device Product Development Tips from experts covering engineering, design and development in Phase One of Development.
-

Explore ways to leverage PCB mounted Microfluidic components for compact, reliable interaction with microfluidic cartridges.
-

Systems engineering can help early-stage biotech companies overcome challenges associated with technology transfer.