Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

The impact of shear stress is critical to effectively design medical devices that handle biological fluids such as proteins or cell culture media. For example, non-physiological shear stress (NPSS) on blood is a key factor because hemolysis (cell rupture) could occur due to accumulated stress.
-

The costs of early-stage medical device development in North America and Europe continue to rise. Increasing technical complexity and the compounding costs of nonclinical and clinical evaluations are driving this trend.
-

Today, 85% of the top 50 healthcare companies use Computational Modeling and Simulation (CM&S) to develop their products and processes. Whether it’s refining overall device parameters or optimizing critical requirements, engineering simulations help reduce development timelines and enhance design exploration.
-

Computational Modeling and Simulation (CM&S) for medical devices has become a pivotal tool across the medical device industry, complementing and often enhancing traditional bench testing and clinical studies.
-

Ariana Wilson and Mark Drlik dive into how the FDA is adopting artificial intelligence to modernize its regulatory processes. With a new chief AI officer in place and rumors of collaboration with OpenAI, the agency is taking major steps to automate review workflows and improve efficiency.
-
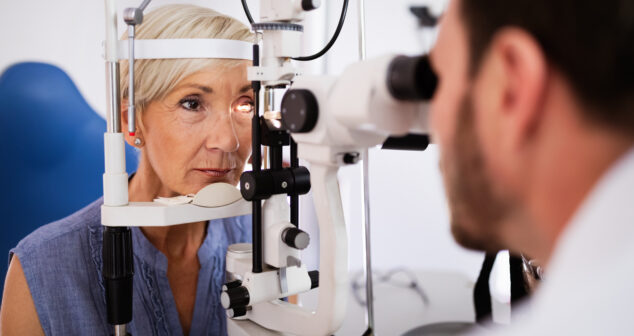
Optics Physicist and Engineer share approaches to performing pre-screen Ophthalmic Instrument Safety Assessment testing in-house.
-

In this episode of MedDevice by Design, Ariana Wilson and Mark Drlik examine what happened, what it means for medical device innovators, and how the FDA’s ASCA (Accreditation Scheme for Conformity Assessment) program helps reduce regulatory risk.
-

We all know medical devices have labels, but how often do we consider their purpose and the effort required to ensure they provide the right information? Device labelling serves as the interface between the manufacturer, the user, and regulatory bodies. (Note that being from Canada, we spell labelling with two Ls.)
-

Sterilization is a critical process in the medical device industry as it provides a reliable way to ensure that devices are free from harmful microorganisms when they are used on patients. This blog talks about the categories of sterilization currently used on medical devices in manufacturing settings. It also addresses concerns surrounding the use of ethylene oxide (EtO), an indispensable method for sterilizing heat and moisture sensitive devices.