Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

Medical device development is a complex process that requires careful attention at every stage.
-

The risk management approach
-
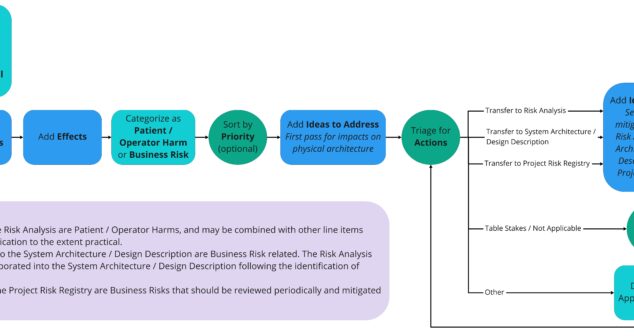
Using xFMEA as a Brainstorming Tool uncovers a powerful early-stage medical device design input tool used successfully at StarFish Medical.
-

PFMEA prevents causes rather than detecting them (or the effects of failures), and should be incorporated into medical device design.
-

Risk management is a key element in your innovation process and an excellent skill. Here are five pro tips to managing innovation risk
-

Engineers, regulatory advisors, and patent filing teams should review the MAUDE database as part of development and due diligence process.
-

Safety implications to choosing lithium batteries in medical devices as well as practical business implications. Avoid these pitfalls.
-

How do we define the quality of a medical device? What attributes cause us to come to the conclusion that a device…