
Why qualitative research for medical device design?
The value of qualitative research in a highly regulated industry
When designing medical devices, it is important to remember that, just like consumer products, the devices we are creating interact with humans. They will be used by humans, on humans. Aspects of design like comfort, perception, behavior, information communication, all play a role in creating this interactive experience. It is safe to say that human behavior and perception is not calculable or quantitative. This requires us to employ qualitative research for medical device design studies and techniques.
Almost a year ago I began my journey into medical device design. I have learned a tremendous amount (and am learning more every day) about the nuances of medical device design and how we approach it. For obvious reasons (and reasons that I very much appreciate) medical device research is heavily focused on the quantitative aspect of the device, such as – Stats, measures, yes or no questions that produce definitive results that cannot be interpreted in any other way. In order to meet regulatory standards and prove efficacy we certainly require hard, calculable data. If a device must deliver 20mL of a specific fluid to a specific area of the human body, we can conduct quantitative research to prove that in fact, it does.
With qualitative research we aim to get to the heart of how someone will interact with the device we are creating. When we understand this, we can shape how someone will (and how well they can) use an object (reducing use error and mitigating risk), the likelihood of them using it (improving uptake), and in some cases, improving the outcome of the object’s intended use. While qualitative research usually does not produce a calculable value that can be hard referenced, it does display trends that can allow us to draw a conclusion.
Here are a few examples that may help to display how a variety of design elements can influence your experience with an object. Some of the following examples fall outside of the medical device realm, but the exact same principles hold true.
Which chair is stronger?

While both of these chairs have the exact same strength and weight rating, there is a good chance that you have a gut reaction to one of these being more durable. If you needed to spec a chair in a high abuse, public area, which would you lean towards?
Which C-Pap nasal mask is more comfortable?

Without having actually tried either of these, there is a very good chance you made an assumption of which device would be more comfortable. An interesting flipside of this example, do you assume that one is more capable or dependable than the other?
Which cordless drill has more torque? Is more durable?

It may surprise you (it certainly did myself) that these two cordless drills have equal torque ratings and spec. If you were on the market for a cordless drill with high torque and good durability, without seeing any sort of spec, which would you choose? I think it is safe to say that one of these communicates power and durability far better than the other.
And finally, which MRI machine is less intimidating to you?
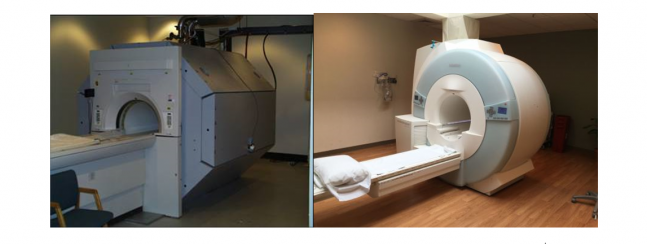
It seems pretty clear which machine would have you a little more at ease before your procedure. As I will speak about in a later article, here we can begin to quantify the importance of qualitative design in the medical industry. In studies from the British Journal of Surgery it has been shown that patients with moderate pre-operative anxiety have an increased probability of complications and increased hospital stays[1].
There is a measured value that describes how user and patient stress can have a direct influence on health procedure outcomes. Through qualitative design research we can inform our industrial design to reduce stress in users and patients and improve outcomes. Thus, a more inviting and approachable device lowers patient anxiety and can result in improved outcomes.
Prior to working with StarFish Medical, I spent 15+ years primarily in consumer product design both freelance and with large product design firms. In that time, I had the pleasure of learning from and working with some renowned and talented ethnographers and behavioral researchers. One of the most important things I learned is probably the simplest, the question of why.
When we understand why someone interacts with an object the way that they do, we can influence how they interact with that object through its design. Getting to the bottom of this allows us to create more enjoyable, accurate and accessible devices. Secondly, it is extremely important for us as designers to remember that we are NOT designing for ourselves; we are designing for the user(s) of the device.
As a 40y/o, Caucasian, middle to upper class male in good health, living in Canada do I really understand the perceptions of a 21 y/o, Asian, low income female living in Australia with diabetes? Plain and simply, no. The only way I or any designer possibly could is through talking to and gaining insight from that individual. That is qualitative research for medical devices. Through gaining this richer understanding of the user we can create truly impactful objects and experiences that have a meaningful impact on lives.
[1] Association between psychological health and wound complications after surgery, British Journal of Surgery, Feb 14, 2017, P. Britteon , N. Cullum, M. Sutton
Mike Loveless is an Industrial Designer at StarFish Medical. Based in Toronto, Mike studied Industrial Design at Humber College and has 15 years experience in consumer product design working both freelance and with full service design firms. Over his next few blogs, he will share some of the tips, tricks and nuanced techniques that he has learned in conducting qualitative user research.