Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

Nick Allan and Nigel Syrotuck explain exactly how aminoglycoside antibiotics work and why they're so effective at killing bacteria.
-

Graphical mind maps created in online whiteboards offer a low-barrier, highly collaborative approach to early risk analysis in medical device development.
-

Early phase concept development is a weird part of a project lifecycle. It is often the most exciting phase, because the team is exploring possibilities, generating new ideas, and turning a fuzzy opportunity into something real.
-
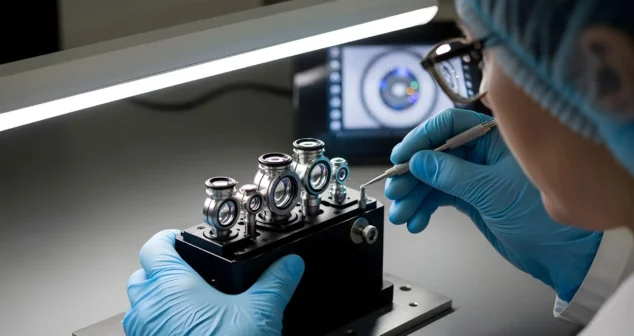
Clinical prototypes must not only function as intended, but also be manufactured, documented, and supported in a way that satisfies regulatory expectations and clinical realities.
-

In this Bio Break episode, Nick and Nigel explore why some GLP-1 drugs are taken daily while others last a full week, and what drives that difference at a molecular level.
-

Choosing the right project estimation approach can make or break a medical device program. Learn when to use a gut feel, rough order of magnitude, or detailed estimate to balance speed, accuracy, and risk.
-

Ozempic is a synthetic version of a hormone your body already makes. That hormone is called GLP-1 (glucagon-like peptide-1), and your intestine releases it naturally when food arrives.
-

Accelerated aging in medical devices is a testing method used to estimate how a product will perform over time by exposing it to elevated conditions, most often heat. In simple terms, it is a way to simulate months or years of aging in a much shorter timeframe.
-

In a recent article for MD+DI, StarFish Medical Software Manager Sean Daniel explores how remote medical devices reliability is becoming a defining challenge as devices move beyond traditional clinical environments into homes, workplaces, and public settings.