Three Physical Connection Methods in Medical Device Test Fixtures
Physical testing is an essential tool for product development, particularly when developing a novel medical device. When well designed, a physical test can accurately represent phenomena that are difficult to model, and physical tests can often be constructed quickly with a minimum of custom components.
Here are three connection methods that I have found useful for physical testing fluid connections in medical device test fixtures. Of the many options, these connection methods are readily available, inexpensive, and well suited for low pressure physical testing. I generally use them for tests involving a small number of test runs, fluids similar to water, and pressures near 1atm.
LUER


Luer ports provide a versatile, inexpensive connection that comes in a wide range of materials and mounting styles. Luer lock is commonly used for medical devices, so it easily integrates with a number of standard parts (tubing, syringes, stopcocks, check valves, etc.). However, their pressure capacity is generally limited (>40psi), and they typically require barbed tube fittings.
When attaching luer fittings to a fixture, it is helpful to anchor the female side of the luer connection. The male side typically has a large grip that is helpful for attaching and removing tubing. We have had success 3D printing female luer ports directly into a test fixture using high res WaterShed XC11122 with build layers normal to the port axis.
PUSH TO CONNECT
More expensive than luer, push to connect fittings provide a quick method of connection with additional pressure capacity, and they do not require a second fitting attached to the tube. They are a useful alternative to barb fittings because 1) they require less force to connect and disconnect, 2) they are less likely to damage the tubing, and 3) they generally keep the flow path equal to the ID of the tubing.

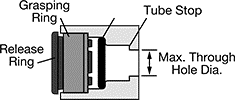
Push to connect fittings also come as inserts that press into a machined hole to create a custom manifold. They can have significantly higher pressure capacity than luer fittings (>200 psi), but they are only compatible with harder tubing (>90A durometer).
O-RING FACE SEAL
Interfacing with small flow channels, an effective O-ring face seal can be made using a circular pocket and a drilled hole. The gland lacks an inner wall to support high external pressure, but it is useful for quickly connecting a standard test fixture to a number of different test articles (e.g. microfluidic cards).
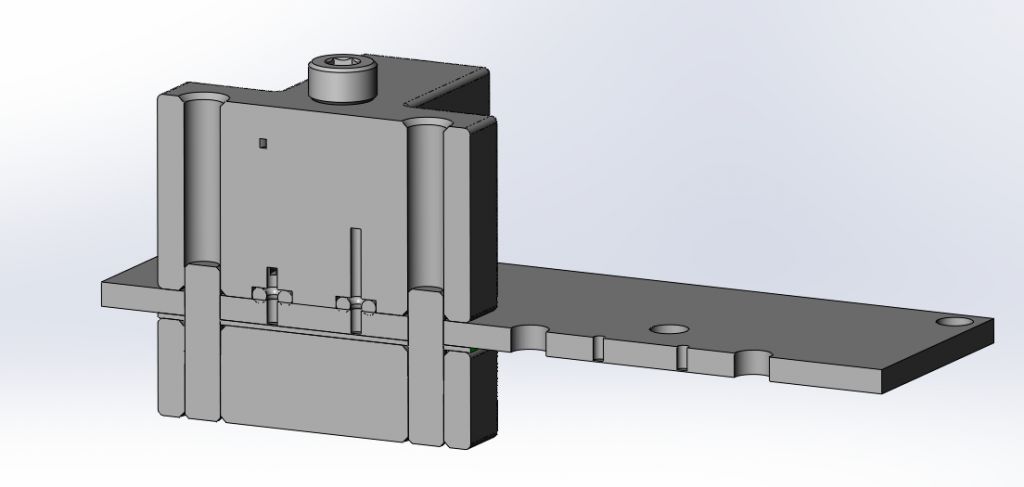
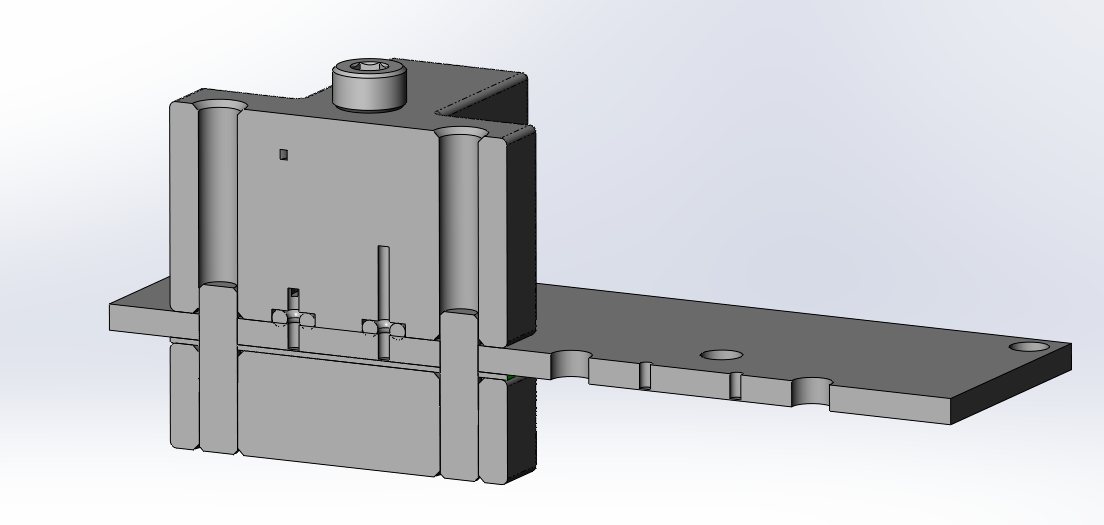
In the image above, we are connected to a microfluidic laminate with two O-ring face seals, locating pins, and a bolted connection. Using a soft (50A durometer) O-ring at low pressure, we have had success using simple face seals with water at low pressures in both machined housings and 3D printed flow manifolds (WaterShed XC11122 with the sealing face on a single build layer, free of support material, and left as printed).
When designing a physical test, or making quick modifications, it is helpful to have a list of options available and some parts on hand. Hopefully, these three ideas for fluid connections in medical device test fixtures are a useful addition to your design tool kit.
Images:
CAD Image: StarFish Medical
Male Luer Fitting: Fisher Scientific
Female Luer Fitting: Cole-Parmer
Push-In Fittings: Tameson
Push-In Fittings in Detail: McMaster
Aaron Philippsen, is a StarFish Medical Mechanical Engineer, and recipient of the company’s Chris Denny Memorial Award for Innovation.
