Optical Detection in Wearable Devices
TL;DR
- Optical detection in wearable devices enables real-time, non-invasive monitoring using light-based sensing.
- Pulse oximetry measures SpO2 and heart rate by analyzing light absorption differences between oxyhemoglobin and deoxyhemoglobin.
- Tissue oxygen monitoring, including SmO2 and rSO2, uses near-infrared spectroscopy to assess oxygen delivery and utilization in muscle and brain tissue.
- The first fluorescence-based optical continuous glucose monitor was FDA cleared in 2024 and uses implantable sensing technology.
- Raman and infrared spectroscopy are emerging approaches that may enable fully non-invasive glucose monitoring in future wearable devices.
- Advances in miniaturization and signal processing are expanding the role of optical biosensing across MedTech wearables.
Introduction
Wearable medical and wellness devices are increasingly commonplace in the healthcare field. For example, devices such as smartwatches incorporate sensors to provide continuous health tracking data to their users.
Many of these sensors use optical (light-based) detection methods. These methods have advantages that make them favorable for wearable devices. They are typically non-invasive, provide specific signals of interest in real- time, and amenable to miniaturization.
In this blog, I discuss how optical detection is used in several common and emerging wearable medical and wellness devices.
Blood Oxygen Saturation (SpO2) and Heart Rate
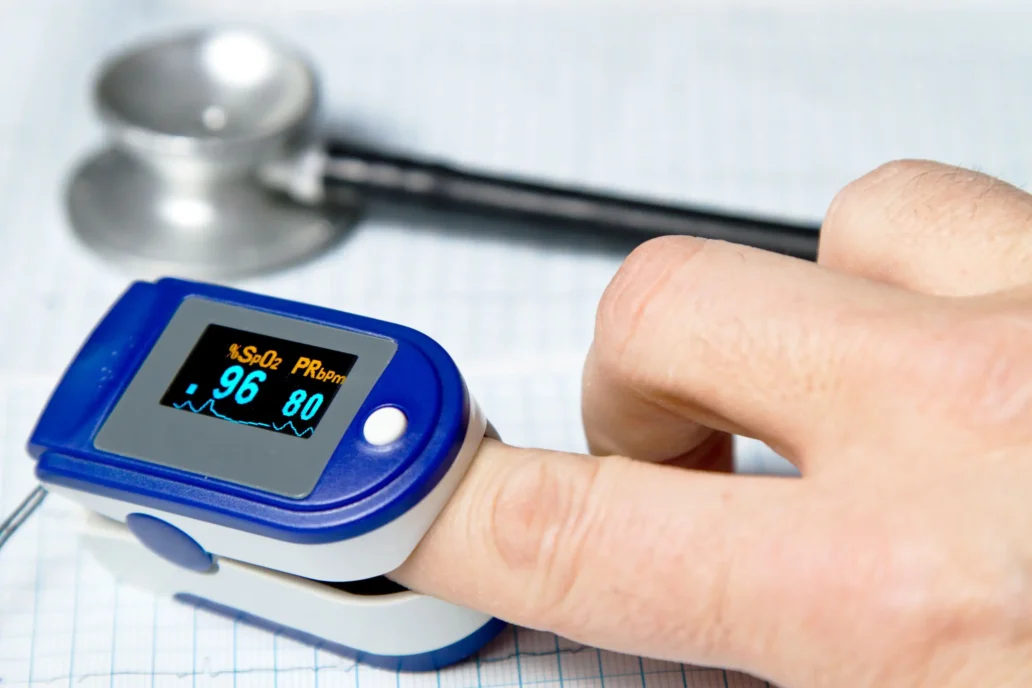
Perhaps the most common use of optical sensors in wearable medical/wellness devices is measuring arterial blood oxygenation (i.e. peripheral capillary oxygen saturation, or SpO2) using pulse oximetry. A person’s heart rate can be measured using the same signal and is often reported alongside SpO2. SpO2 monitors are increasingly incorporated into smartwatches and similar devices.
Oxygen saturation of blood corresponds to the fraction of hemoglobin in the blood that binds an oxygen molecule for transport, sometimes called “oxyhemoglobin.” Hemoglobin that is not bound to an oxygen molecule is called ’deoxyhemoglobin.” To find this fraction, light at two different wavelength is reflected from or transmitted though tissue. A red (usually ~660 nm) light source is used to differentiate oxyhemoglobin from deoxyhemoglobin, as deoxyhemoglobin absorbs more strongly at that wavelength. A light source at an infrared (IR) wavelength (usually ~940 nm), where the two types of hemoglobin absorb similarly, is used as a blood volume reference[1]. As the heart pumps blood, the volume of oxygenated arterial blood at the sensor pulses. By looking at changes in absorption at the two wavelengths in the generated pulsatile signal, the oxygen saturation of arterial blood can be determined.[2].

The main purpose of medical SpO2 monitors is to ensure that there is sufficient oxygen in the blood for tissues in the body to function normally. Continuous SpO2 monitoring is common for patients in hospitals and may be used for patients receiving anesthesia [1], patients with respiratory issues [2], and patients with acute heart conditions [3]. Home spot checks or continuous monitoring may be prescribed for evaluation of sleep apnea (overnight), monitoring chronic obstructive pulmonary disease, and for patients requiring supplemental oxygen at home. In wellness devices, SpO2 monitoring is generally less accurate and is primarily used for general health awareness and to track trends during fitness, sleep, or travel to high altitudes.
Medical pulse oximeters are usually worn on the fingertip. Some popular medically-cleared pulse oximeters include the Nellcor portable SpO₂ patient monitor, Masimo MightySat RX, and Nonin 8500. Pulse oximeters/heart rate monitors are also incorporated into many popular wellness devices. These are more commonly worn on the wrist or as a ring. Examples include certain models of the Apple Watch, Samsung Galaxy Watch, and the Oura Ring.
Tissue Oxygen Saturation (SmO2, rSO2)
A similar use of optics in wearables is the measurement of local muscle oxygen saturation (SmO2), especially in specific tissues such as the brain (regional tissue oxygen saturation, or rSO2). In contrast to SpO2 monitors, which specifically measure oxygenated arterial blood that has largely not yet delivered oxygen to tissue, SmO2 monitoring queries the oxygenation of all blood in a volume of muscle near the sensor.
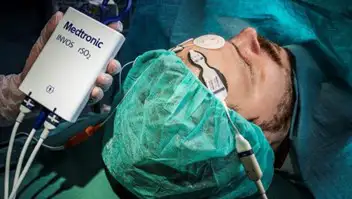
SmO2 monitoring is often used in sports, fitness, and rehabilitation to track muscle activity. The SmO2 signal gives information on the delivery and use of oxygen to the muscle tissues. If SmO2 falls too low during activity, athletes/patients may decide to reduce the intensity of their workout. Regional tissue oxygen saturation monitoring is clinically applied to monitor brain oxygenation during various surgeries [4], blood perfusion in transplanted tissue [5], and early signs of tissue hypoxia in intensive care unit (ICU) settings [6].
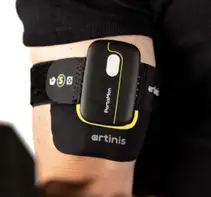
SmO2/rSO2 monitoring measures the relative attenuation of light at two (or more) wavelengths and uses the different attenuation of oxy- and deoxyhemoglobin (and sometimes myoglobin) at those wavelengths to determine the relative concentration of the oxygenated species. Since the intent is to measure the oxygenation of all blood in the volume, the pulsatile signal that isolates oxygenated arterial blood in SpO2 measurement is not required. Instead, light at the two wavelengths is sent into the tissue, with detectors placed at a distance from the emitter on the same side of the tissue. Some of the light diffusely reflects through the tissue, reaching the detectors. The known extinctions of the two hemoglobin species and the effective path length between the emitter and detectors are used to infer oxygen saturation in the queried tissue from the relative intensities of the different wavelengths reaching the detector. The wavelengths used are typically in the infrared region (usually between 700 nm and 900 nm), where the light penetrates relatively deep into tissue but is absorbed by the relevant molecules in blood. The measurement performed is a variation of near-infrared spectroscopy (NIRS).
Clinical rSO2 systems are typically not “wearables” but nonetheless include sensors that are worn on the body during measurement to provide real-time feedback. These systems include the Medtronic INVOS systems and the Massimo O3. Fully wearable SmO2 monitors are available for sports, fitness, and research. Examples include the Moxy Monitor and the Artinis PortaMon.
Continuous Glucose Monitors (CGMs)
Continuous glucose monitors (CGMs) are wearable devices that track blood sugar in realtime and are mostly used for management of diabetes. The overwhelming majority of CGMs do not use optical detection (instead relying on electrochemistry), but several methods are emerging that could make optical CGMs more common.

In 2024, the first fluorescence-based CGM, the Senseonics Eversense 365, was cleared for use by the US FDA [7]. A small sensor is implanted under the skin and transmits data to a removable wearable device that is placed over the sensor. The sensor contains a hydrogel that contains molecules that reversibly bind to glucose and are fluorescent when bound. A light source in the sensor excites the bound molecules, which then emit fluoresced light proportional to the concentration of glucose in the interstitial fluid. Additionally, a second hydrogel contains a molecule that reacts chemically with reactive oxygen species, producing a fluorescence signal that is correlated with oxidation of the glucose-detecting fluorescent molecules. This second fluorescence measurement is used to adjust the measured glucose level to account for changes in oxidation of the sensor over time. A result of this adjustment is that this CGM is claimed to last much longer than typical CGMs (one year versus ~two weeks).
Two emerging technologies that may enable optical wearable CGMs in the future are Raman spectroscopy and IR spectroscopy-based glucose detection. Both methods probe the characteristic vibrational modes of molecules in the interstitial fluid and look for vibrational frequencies characteristic of glucose. The main advantage of these methods is that they are non-invasive; contrary to existing CGMs, nothing is required to pierce or be implanted in the skin.
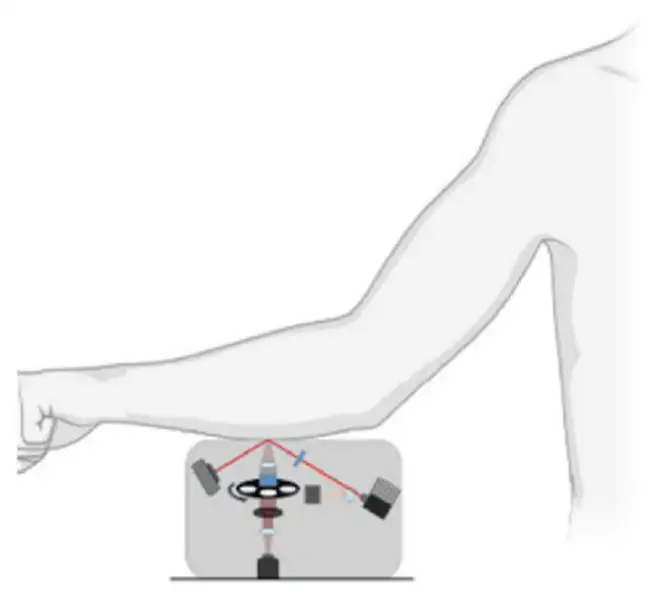
Raman spectroscopy-based methods look at light scattered from tissue to measure glucose levels. A laser is shined on the skin and the light scattered from the tissue is collected and dispersed and/or filtered so that the constituent wavelengths of the returned light can be analyzed. Most of the scattered light will be at the same wavelength as the incident laser, but a small amount will scatter with measurable differences in energy (i.e. wavelength) corresponding to the amounts of energy needed to excite certain vibrational modes of molecules in the tissue. Vibrational energies characteristic of glucose are already known; the presence and magnitude of corresponding energy shifts in the scattered light can be used to infer blood glucose levels. IR spectroscopy-based methods, in contrast, lookfor absorption of IR light at the specific energies (i.e. wavelengths) of light needed to excite glucose’s vibrational modes. The tissue is illuminated with IR over a range of wavelengths containing light capable of exciting glucose’s vibrational modes. Light reflected from or transmitted through the sample is then analyzed, and the magnitude of light lost to absorption at the characteristic wavelengths is observed.
Both Raman and IR-based techniques are currently in research, with some clinical studies but no commercially released or medically cleared devices available yet. Challenges include the signal strength (weak in both cases), confounding signals from tissue and water (requiring sophisticated data analysis), and the relative complexity of the optical setup. Raman-based methods have shown progress towardminiaturization [8] [9], with the ultimate goal of making a wearable device [10]. If successful, devices based on these technologies could provide truly non-invasive CGMs in the future.
Conclusion
Optical detection is a key technology in wearable medical and wellness devices. Pulse oximeters are ubiquitous in clinical settings, and their consumer-wearable counterparts (such as the corresponding modules on smartwatches) are growing in popularity. Tissue oxygenation monitors have seen clinical use in monitoring cerebral or tissue hypoxia, and there are multiple examples of consumer models typically used for sports and fitness. The first wearable optical CGM was released in 2024, with promising developments showing the possibility of non-invasive optical CGMs on the horizon. With further development, optical detection modalities have the potential to further expand the suite of real-time biological signals that can be monitored in non-invasive wearable devices.
Ryan Field is an Optical Engineer at StarFish Medical. Ryan holds a PhD in Physics from the University of Toronto. As a post doctoral fellow, he worked on the development of high-power picosecond infrared laser systems for surgical applications as well as a spectrometer from home materials.
Footnotes
[1] In some cases, a green LED is used to measure heart rate (though that is not generally required in SpO2 monitors, as heart rate can be measured from the pulsatile red and IR signals).
[2] For a more detailed explanation of how pulse oximetry works, see Brian King’s blog at https://starfishmedical.com/resource/how-pulse-oximeters-work/
References
[1] American Society of Anesthesiologists, “Standards for Basic Anesthetic Monitoring,” [Online]. Available: https://www.asahq.org/standards-and-practice-parameters/standards-for-basic-anesthetic-monitoring. [Accessed 23 February 2026].
[2] K. Torp, P. Modi, E. J. Pollard and L. V. Simon, “Pulse Oximetry – StatPearls – NCBI Bookshelf,” [Online]. Available: https://www.ncbi.nlm.nih.gov/books/NBK470348/. [Accessed 23 February 2026].
[3] J. Masip, M. Gaya, J. Paez, A. Betbese, F. Vecilla, R. Manresa and P. Ruiz, “Pulse Oximetry in the Diagnosis of Acute Heart Failure,” Rev. Esp. Cardiol., vol. 65, no. 10, pp. 879-884, 2012.
[4] J. Qin, G. Wang, D. Gu, J. Li, J. Zhang, M. Ge, X. He and X. Ma, “The impact of cerebral oxygen saturation monitoring on perioperative neurocognitive disorders: a meta-analysis and economic analysis,” Front. Med., vol. 13, no. 1677218, pp. 1-21, 2026.
[5] Z. Cai, J. Zhang, J. Zhang, F. Zhao, G. Yu, Y. Li and H. Ding, “Evaluation of near infrared spectroscopy in monitoring postoperative regional tissue oxygen saturation for fibular flaps,” J. Plast. Reconstr. Aesthet. Surg., vol. 3, no. 289-296, p. 61, 2008.
[6] R. Imai, T. Abe, K. Iwata, S. Yamaguchi, T. Kitai and A. Tsubaki, “Regional cerebral oxygen saturation during initial mobilization of critically ill patients is associated with clinical outcomes: a prospective observational study,” Intensive Care Medicine Experimental, vol. 13, no. 13, pp. 1-14, 2025.
[7] Senseonics, “Eversense 365 Receives FDA Clearance: The World’s First One Year CGM,” [Online]. Available: https://www.senseonics.com/investor-relations/news-releases/2024/09-17-2024-120118174. [Accessed 23 February 2026].
[8] A. Bresci, Y. Kim, M. Jue, P. T. C. So and J. K. Kang, “Band-Pass Raman Spectroscopy Unlocks Compact Point-of-Care Noninvasive Continuous Glucose Monitoring,” Anal. Chem., vol. 97, pp. 27020-27026, 2025.
[9] L. Liu, K. Zhan, J. Kilpijärvi, M. Kinnunen, Y. Zhao, Y. Zhang, M. W. Yaltaye, Y. Lu, A. Zhyvolozhnyi, A. Samoylenko, S. Vainio and J. Huang, “Portable and label-free optical detection of sweat glucose using functionalized plasmonic nanopillar array,” Microsystems & Nanoengineering, vol. 12, no. 43, pp. 1-11, 2026.
[10] A. Booth, “Raman spectroscopy enables noninvasive blood glucose monitoring in compact MIT device,” [Online]. Available: https://www.labmate-online.com/news/raman/135/breaking-news/raman-spectroscopy-enables-noninvasive-blood-glucose-monitoring-in-compact-mit-device/66340. [Accessed 23 February 2026].
Image Credits
Figure 1: Adobe Stock & https://helios-i.mashable.com/imagery/reviews/007PyrbfR6ywbelJSTcnEUL/images-19.fill.size_2000x1334.v1623361899.jpg
Figure 3: https://www.artinis.com/portamon-mkiii
Figure 4: https://www.eversensecgm.com/siteassets/eversense-365/transmitter-min.png
Figure 5: A. Bresci, Y. Kim, M. Jue, P. T. C. So and J. K. Kang, “Band-Pass Raman Spectroscopy Unlocks Compact Point-of-Care Noninvasive Continuous Glucose Monitoring,” Anal. Chem., vol. 97, pp. 27020-27026, 2025.
Related Resources
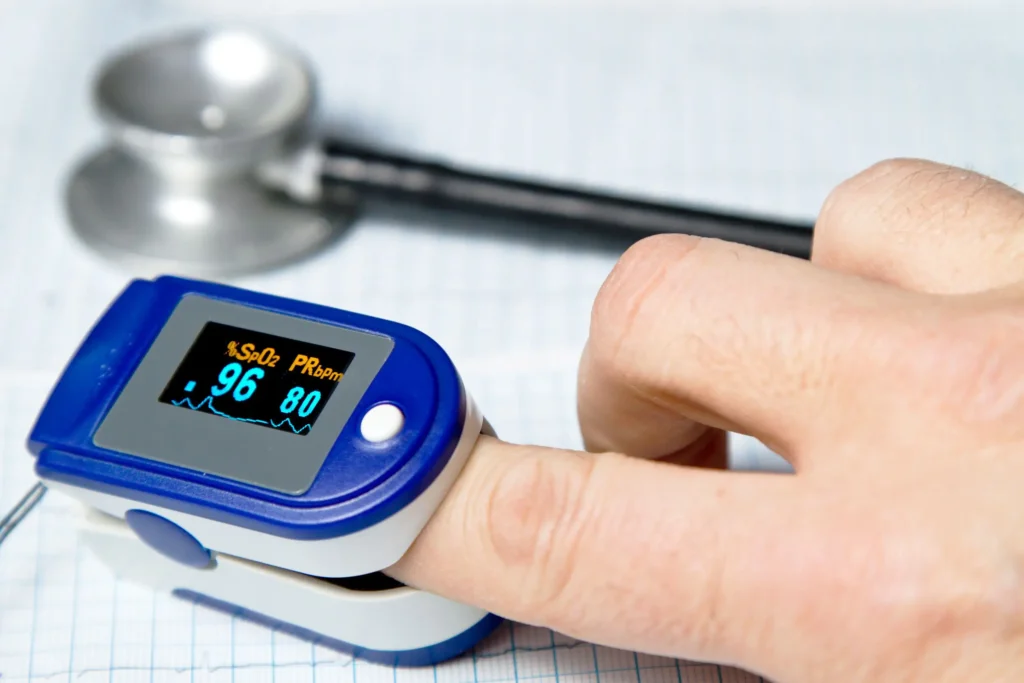
Wearable medical and wellness devices are increasingly commonplace in the healthcare field. For example, devices such as smartwatches incorporate sensors to provide continuous health tracking data to their users.
Commonalities and points of potential confusion of the “big 4” incoherent-light hazard classification and standards for medical devices.

Microfluidic Optics explores ways that lenses can be implemented on microfluidic cartridges can play an important role in microfluidic platforms,
Medical device color management overview of why colour-rendering accuracy is critical for LEDs CRIs and Full-Colour Imaging.