Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-
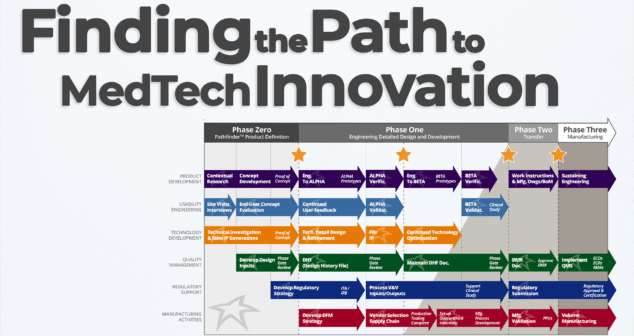
Nick and Joris explore one of the most dynamic early-phase services at StarFish Medical: the Pathfinder Program. If you're a medtech innovator with a promising concept or prototype, Pathfinder helps you identify the right path forward—before you invest millions in development.
-
Optics Physicist and Engineer share approaches to performing pre-screen Ophthalmic Instrument Safety Assessment testing in-house.
-

If you’ve ever been to the hospital, you’ll know that one of the first things hospital staff do is attach “that finger clip device” to your finger. “That device” is called a Pulse Oximeter, and it provides information on pulse rate and blood oxygenation.
-
Fluorescence Imaging in Medical Devices outlines medical applications and examples of devices that use fluorescence for imaging.
-

Computer Vision for Medical Devices is constantly evolving and incorporating new techniques and technologies as they emerge.
-
With the recent developments and seemingly ubiquitous nature of real time glucose monitoring and availability of smart wearable tech, the development of a theranostic band-aid seems inevitable. But how practical would this be? Is there a Theranostic wound dressings market?
-

In this episode of MedDevice by Design, Ariana Wilson and Mark Drlik examine what happened, what it means for medical device innovators, and how the FDA’s ASCA (Accreditation Scheme for Conformity Assessment) program helps reduce regulatory risk.
-

In this episode of Bio Break, Nick Allan and Joris van der Heijden explore a critical but often overlooked topic in healthcare innovation: prevention. While most conversations about medical devices revolve around treatment, the duo shifts the focus to technologies that help people avoid hospitalization altogether. Preventive medical devices and diagnostic tools are quietly transforming healthcare by catching diseases earlier and reducing the need for invasive procedures.
-

Ariana Wilson and Mark Drlik break down a powerful visual framework for understanding what makes a medtech product, and the company behind it, truly successful. Using a triple Venn diagram, Mark explains how strategic alignment across feasibility, viability, and desirability can drive better product outcomes and business success in the medical device industry.