Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

Eight robust First-to-Market commercialization tips and insights addressing trade-offs when bringing a medical device to market.
-
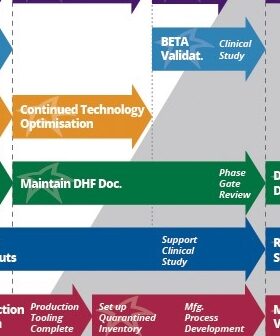
Why build a beta medical device? After a medical device receives regulatory clearance, it’s fairly burdensome to make changes. Beta device benefits are explained by experts.
-

Medical Device Playbook was conceived to inspire entrepreneurship and connect the many players. Here are commercialization Insights from attending the event.
-

Medical device development experts share their biggest medical commercialization lessons learned over 20 years.
-

9 medical device commercialization resolutions recommended by our team of engineers, designers, QA/RA, and manufacturing professionals.
-

Any medical device first to market production release strategy that minimizes time requires an honest examination of the phase-gate process.
-

MaRS EXCITE collaborates with the Ministry of Health to support adoption and diffusion of promising technologies within Ontario.
-
Even in medical device design every product has a story, and like most good stories it is how it overcame failure.
-

Joel Weinstein is the real deal medical device serial entrepreneur. Joel spoke at many Medical Device Playbooks starting with Toronto 2016.