Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

Jason McGee explores how the Rightness Trap Medtech dynamic can quietly erode enterprise value in medical device companies.
-

Scott Phillips, CEO of StarFish Medical, sits down with Peter van der Velden, Managing General Partner at Lumira Ventures, to explore the strategic thinking behind major MedTech transactions and investments.
-

Every MedTech startup begins with a hypothesis, an idea that could transform patient outcomes, simplify delivery of care, or improve how clinicians diagnose and treat patients.
-

In Medtech, a successful exit isn’t just about having an innovative device, it’s about building a business that potential buyers and investors can clearly see a future in.
-

Nick Allan and Nigel Syrotuck dive into the evolution of patent documentation habits in engineering and medical device development.
-
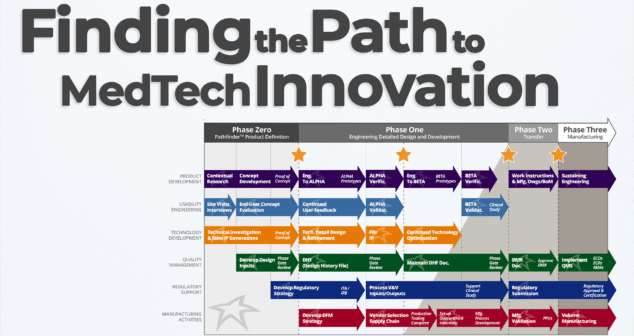
Nick and Joris explore one of the most dynamic early-phase services at StarFish Medical: the Pathfinder Program. If you're a medtech innovator with a promising concept or prototype, Pathfinder helps you identify the right path forward—before you invest millions in development.
-

Ariana Wilson and Mark Drlik break down a powerful visual framework for understanding what makes a medtech product, and the company behind it, truly successful. Using a triple Venn diagram, Mark explains how strategic alignment across feasibility, viability, and desirability can drive better product outcomes and business success in the medical device industry.
-

What are the real costs of developing a medical device? In this episode of Bio Break, Nick and Joris dive into one of the most frequently asked questions they hear from clients: How much does it cost to develop a medical device?
-

Nick and Joris explore the fascinating world of repurposing existing medical device technologies for new market sectors. As engineers and innovators, we often focus on creating brand-new solutions, but what about leveraging tried-and-true technologies to expand into untapped markets? This strategy not only opens doors to new revenue streams but also maximizes the potential of existing innovations.