Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

Case studies show value of qualitative research in medical device design, illustrate human behavior is not calculable or quantitative.
-
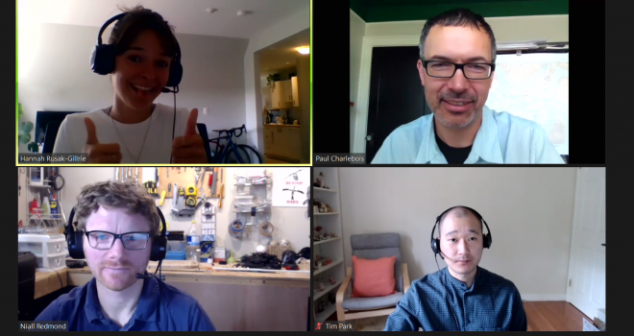
COVID-19 Ventilator Design team discuss their top insights and lessons learned from the time constrained, critical ventilator project.
-

Formative Evaluations and Tests details the differences between these two methods, and the appropriate circumstances for their application.
-

Creative Human Factors Processes for COVID-19 solutions to combat logistical hurdles for human factors research and evaluation activities.
-

Less is More When Designing Medical Device user interfaces, device enclosures, and instructions. Four steps to success!
-

There are a lot of things to consider when creating a summative usability test. The first time can be overwhelming. What scenarios do I test? How do I identify a critical task? How many participants do I need? Do I need to test the training? In this eGuide I will help you avoid common mistakes, like using a formative test structure for a summative usability test. I will also provide some tricks and tips to help you get the most out of your test participants and testing.
-
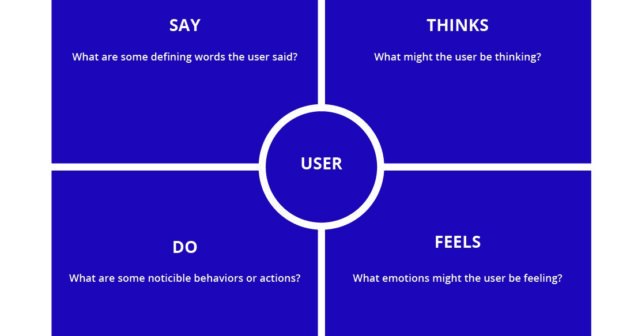
Empathic Design paying attention to what female users say, think, do, and feel, gives a holistic understanding of user desires and needs.
-

Alarm fatigue is influenced by a variety of factors. End user and patient behaviours are most identifiable through procedural observation.
-

Setting up observation sessions can be quite time consuming. Three tips to make the most of your time and results.