Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-
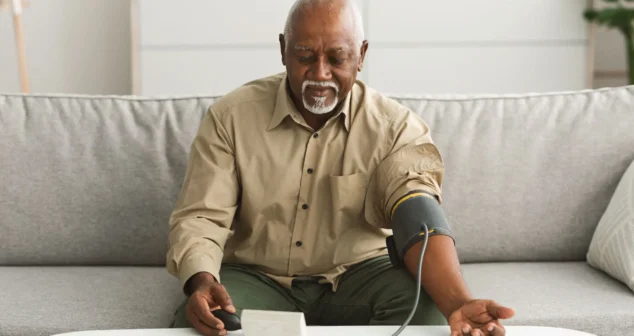
Most medical devices were designed for clinical settings, not the patients and caregivers who increasingly rely on them at home. Here's what good home-use device design actually requires.
-

How do you measure comfort in medical device design? Explore the tools, scales, and study design principles that turn a subjective experience into actionable design data.
-

Gathering health data has enormous value for spotting risks, improving care, and advancing science. The problem isn't capturing the data. The problem is how we choose to present it and who we're really serving when we do.
-
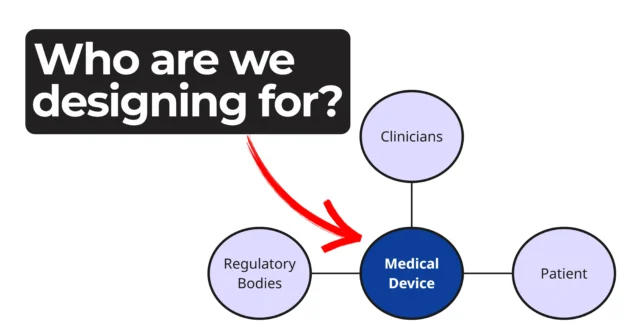
Every phase of a device’s life cycle involves different people with distinct needs—from clinicians and patients to service technicians and regulatory bodies.
-

Nigel Syrotuck and Nick Allan explore the surprising reality of inhaler spacer use. While these devices are often thought of as tools for children with asthma, research shows that adults struggle with them too.
-

In this Before the Build episode, Eric Olson and Paul Charlebois dive into the importance of organ transplant logistics when designing effective medical devices.
-
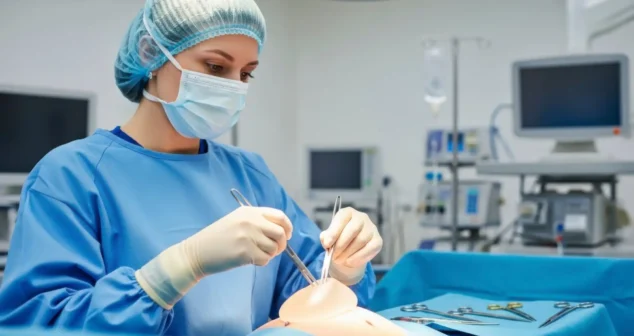
The simplest and least expensive way to train users of medical devices is to ask them study the Instructions for Use (IFU) beforehand.
-

In the complex world of medical device development, creating effective prototypes is more than just a creative exercise, it’s a regulatory necessity and a strategic imperative.
-

User Experience (UX), Human Factors (HF), and Industrial Design (ID) each have a major impact on the success of new medical devices. Their influence is especially important during product definition and early phases of device development.