Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-

In this episode of Bio Break, Nick and Joris dive into the fascinating realm of biosensors, showcasing how nature’s biological processes inspire groundbreaking innovations in medical device technology. From jellyfish to fireflies, the natural world has provided invaluable tools that are transforming diagnostics and research.
-

Dive into the world of assay development with this informative episode of Bio Break, where Nick and Joris explore two critical concepts: Limit of Detection (LOD) and Limit of Quantification (LOQ). These terms might sound similar, but their implications for clinical diagnostics and medical devices are vastly different. Whether you’re an engineer, researcher, or product developer, this video sheds light on why both LOD and LOQ are vital in ensuring the precision and reliability of diagnostic tests.
-

In this episode of Bio Break, Nick and Joris dive into a common challenge faced by researchers and product developers: the use of pipettes in laboratory workflows and the need for alternatives in non-laboratory environments. From the precision of the trusty P200 pipette to innovative solutions like microfluidics, this discussion highlights the strengths and limitations of fluid handling tools in research and beyond.
-
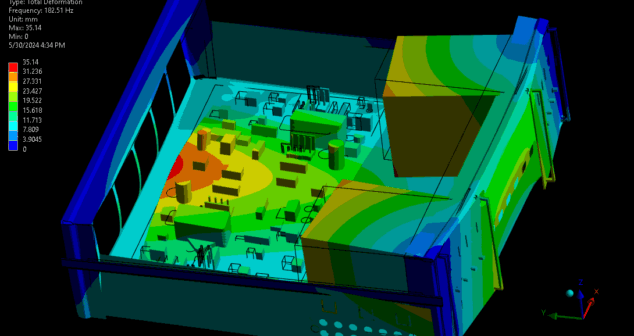
The impact of vibration on medical devices can be understood through a combination of vibration analysis and testing. This article reviews medical devices most susceptible to vibration, provides basic theory and methods of vibration analysis, and summarizes the benefits of using modal analysis when developing medical devices.
-

Opening a biomed lab includes managing multi-layer organizational involvement all the way from facilities to the head of operations. When starting a new lab from scratch, the first step is to fully understand the organization’s mission and business goals. This will impact how you approach setup and any purchasing decisions you make. This article describes 12 essential steps to set up a biomed lab.
-

In this episode of Bio Break, Joris van der Heijden and Nick Allan discuss the critical role of specialized facilities in the successful development of medical devices. From testing laboratories to clean rooms, they explore the infrastructure needed to support innovation and ensure safe, effective products reach the market.
-

Tips for building a medical device optical breadboard prototype. When designing a medical device made of complex, multi-functional mutually interacting subsystems it’s best to de-risk those subsystems instead of coming up with a paper design, assembling it, and hoping it works first time. De-risking individual subsystem behaviours one at a time enables more-focused assessments and improves troubleshooting.
-
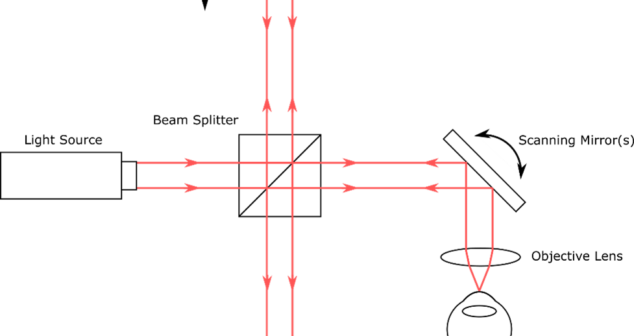
Overview of Optical Coherence Tomography (OCT) Types includes some advantages and disadvantages of the imaging techniques in medical devices.
-

Medical Device Design for Testability during development explores the pros and cons of its application throughout the entire design process design concern throughout the whole design process, ensuring that all parts of the product can be both manipulated and monitored allowing for thorough testing.