Bacterial Endotoxin Testing Explained
Bacterial endotoxin testing plays a critical role in medical device safety. In this Bio Break episode, Nick and Nigel take a surprising path from aliens and crabs to one of the most important quality checks in MedTech.
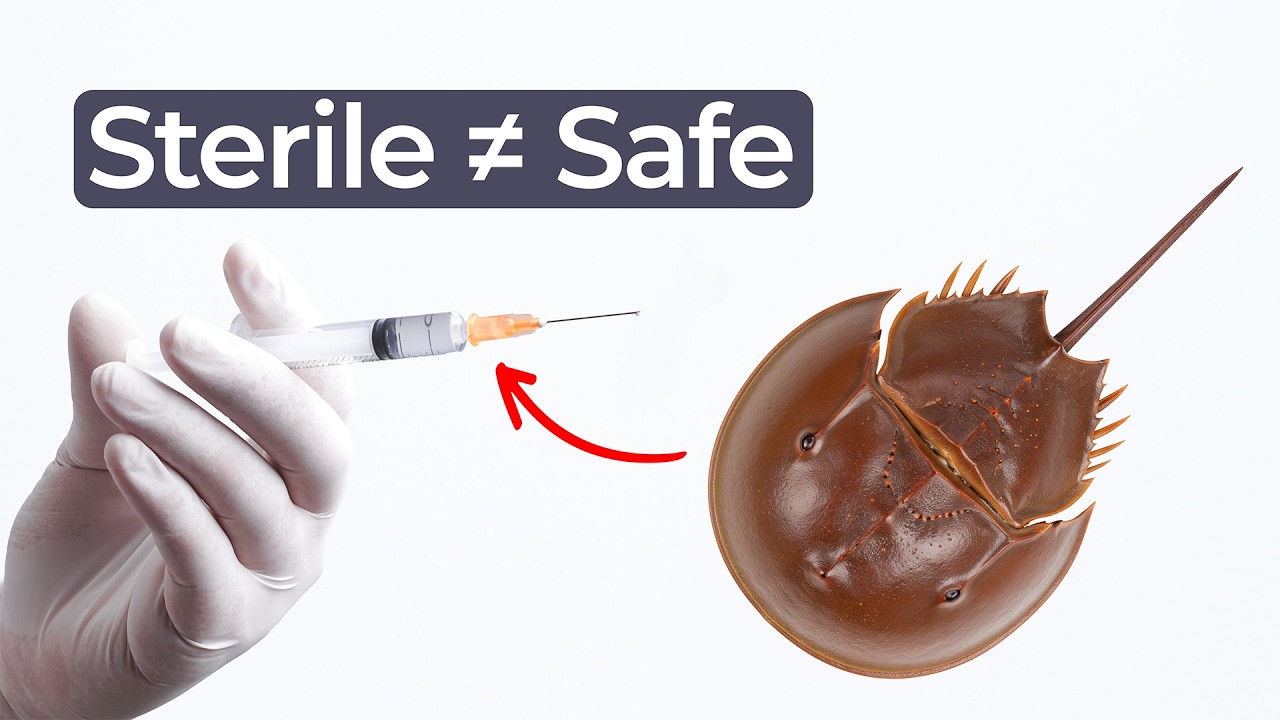
Although the conversation starts with speculation about what aliens might look like, it quickly turns to evolution, horseshoe crabs, and a remarkable property found in their blood. That property forms the foundation of the LAL test, which is widely used to detect endotoxins on medical devices.
From Horseshoe Crabs to the LAL Test
Horseshoe crabs have a unique immune response. When their blood encounters gram negative bacteria, it forms a precipitate that helps neutralize the threat. This reaction is incredibly sensitive.
Scientists harnessed this natural defense mechanism to develop the LAL test. By extracting horseshoe crab blood, laboratories can detect extremely small amounts of endotoxin contamination. As a result, the assay is now a standard method for endotoxin detection in medical devices.
The LAL test does not look for live bacteria. Instead, it detects lipopolysaccharide, which is part of the outer membrane of gram negative bacteria. You can think of it as the “skin” of the bacteria. Even after sterilization, that outer membrane may remain.
Why Bacterial Endotoxin Testing Matters for Medical Devices
Bacterial endotoxin testing is essential because endotoxins can trigger serious immune responses in patients. Our immune systems are primed to detect these molecules. When endotoxins enter the body, they can cause fever and other inflammatory reactions. These substances are known as pyrogens because they generate fever.
Importantly, sterilization alone does not eliminate endotoxins. Radiation may kill live bacteria on a syringe, for example, but the bacterial remnants can remain. Therefore, a device can be sterile yet still contain endotoxins.
That is why endotoxin detection is part of post-sterilization assessment. It verifies not only sterility, but also cleanliness and overall safety.
A Reminder of Biology’s Role in MedTech
This episode highlights how natural biology often informs medical technology. An ancient marine species provides the foundation for a modern safety test used across the industry.
It also reinforces a key lesson in MedTech development. Sterility is not the whole story. Leftover biological components can still create risk. Therefore, careful testing remains critical before a device reaches patients.
Enjoying Bio Break? Sign up to get new episodes sent to your inbox.
Related Resources

This medical vs wellness example shows how device classification can directly change functionality. Even when hardware is similar, what the device is allowed to do can be very different.

Modern medical devices are no longer confined to hospital settings. Wearable cardiac monitors, home respiratory systems, and remote patient monitoring devices now operate within broader digital health networks.

This blog provides an overview of the major steps involved in estimating cost and schedule. Though this is specifically based on medical devices, similar projects across a variety of industries will follow similar steps.

Medical device teams developing embedded and cross-platform GUIs can accelerate delivery without compromising usability or validation by choosing the right framework early and designing for performance, portability, and maintainability.