Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-
Nick and Nigel dive into the world of jet injector drug delivery. This needle-free method, made popular in science fiction and real-world vaccines, is still used today.
-
Nick and Nigel explore a surprising approach to injection pain reduction using suction technology. What started as an unusual product order at StarFish Medical led to important insights on improving patient comfort during injections.
-

Nigel and Nick explore microneedle drug delivery—a growing field in medtech that aims to improve patient comfort and treatment compliance. While the term "microneedles" may sound futuristic, this technology has been around for years.
-

Nick and Nigel discuss an often-overlooked but increasingly relevant form of drug delivery: suppositories. While typically considered old-fashioned, suppositories are experiencing a resurgence in modern MedTech thanks to their versatility, systemic absorption benefits, and emerging formulation technologies.
-

While most people think of Botox as a simple beauty treatment, there’s a surprising amount of engineering, anatomy, and precision behind the process.
-

A theranostic wound dressing does more than cover a cut—it actively detects infection and delivers targeted treatment.
-

Nick and Joris break down what a DHF is, why it's required, and how it plays a vital role throughout the development lifecycle.
-

Nick Allan and Joris van der Heijden revisit one of StarFish Medical’s most successful Pathfinder journeys, showcasing how a bold research concept evolved into a fully realized clinical diagnostic device.
-
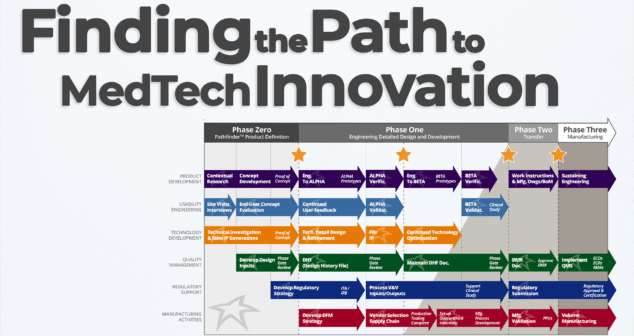
Nick and Joris explore one of the most dynamic early-phase services at StarFish Medical: the Pathfinder Program. If you're a medtech innovator with a promising concept or prototype, Pathfinder helps you identify the right path forward—before you invest millions in development.