Resource Centre
Discover a wealth of knowledge and insights from the experts at StarFish Medical. Our Resource Centre offers product development tips, reviews of new and cutting-edge technologies, and in-depth articles on regulatory updates and compliance in medical device development.
-
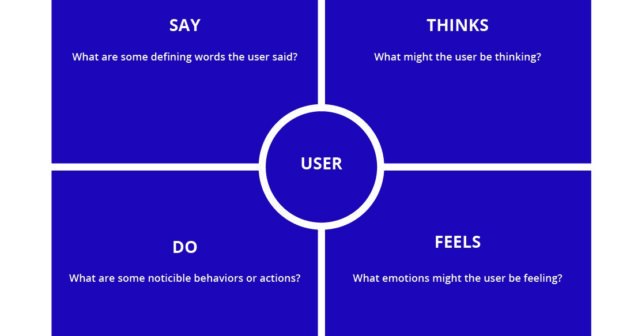
Empathic Design paying attention to what female users say, think, do, and feel, gives a holistic understanding of user desires and needs.
-

Alarm fatigue is influenced by a variety of factors. End user and patient behaviours are most identifiable through procedural observation.
-

Setting up observation sessions can be quite time consuming. Three tips to make the most of your time and results.
-

When observing a summative evaluation it is crucial that the observers do not influence the participants. Tips to avoid influencing results.
-

MaRS EXCITE collaborates with the Ministry of Health to support adoption and diffusion of promising technologies within Ontario.
-

Proper documentation is an up-front investment of time and effort that pays big dividends later in medical device development.
-

6 benefits mean the FDA program for breakthrough medical devices may be your best regulatory option for novel medical devices.
-

FDA's Early Feasibility Studies Program (EFS) is a great option in early stage development looking to advance your program by gathering data.
-

Proper research and communication with authorities in each country of interest will make Medical Device International Regulations smoother to navigate.